1. Introduction
The need for energy among people is growing as science and technology continue to improve. Energy use, however, also results in massive emissions of greenhouse gases that pose a serious threat to the natural balance of the planet. In 1996, Nobel laureate in Chemistry, Richard Errett Smalley, identified energy and environmental issues as one of the top ten crises facing humanity in the next 50 years. In September 2020, the Chinese government, for the first time, proposed the goal of peaking carbon emissions before 2030 and striving for carbon neutrality by 2060, demonstrating China's determination to address carbon emissions. As a major emitter. China accounts for approximately 30% of the world's total emissions, with frequent news reports highlighting the environmental pollution caused by traditional fossil fuels such as coal and oil [1]. In order to alleviate environmental pressure and achieve the goals of carbon peaking and carbon neutrality, it is imperative to develop new energy storage technologies. LIBs, as a mature energy storage technology, have reduced our reliance on fossil fuels over the past few decades.
However, Li resources are few and unevenly distributed, which presents problems. Li abundance in the Earth's crust is roughly 0.0065%, as shown in Figure 1, which leads to high prices for LIBs. Additionally, the extraction of Li minerals results in severe environmental degradation. The development of battery energy storage technologies aims to reduce environmental damage. In contrast, Li is three orders of magnitude more prevalent than Na, which is present in the Earth's crust in abundance at about 2.73% [2,3]. Seawater contains large amounts of Na, making it a more accessible and sustainable supply. Additionally, Na belongs to the same group as Li, exhibiting similar physical and chemical properties, making it more suitable as a new generation of energy storage technology.
Currently, SIBs are gradually transitioning from the laboratory to industrialization. As early as July 2021, CATL released the first generation of SIBs, with a single-cell energy density reaching 160 Wh/kg, the highest level globally [4]. In April 2023, CATL officially announced that SIBs will be first deployed in Chery vehicles, with the potential to replace LIBs in larger-scale commercial production. This paper primarily explores common cathode materials for LIBs on the market, such as lithium iron phosphate (LFP), nickel cobalt manganese (NCM), and lithium cobalt oxide (LCO) [5,6]. It focuses on three representative cathode materials for SIBs, namely layered transition metal oxides, polyanionic compounds, and Prussian blue analogs [2,7-9], and discusses their current research progress.
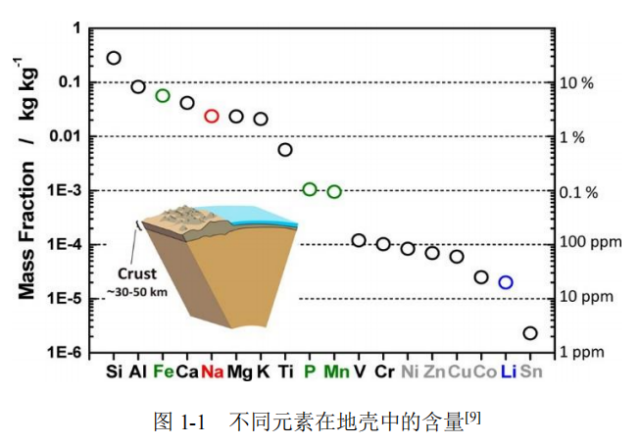
Figure 1. Elemental richness in the crust of the Earth [10].
2. Lithium-ion battery
2.1. Working principle
A LIB is a device that converts chemical energy into electrical energy and usually consists of a cathode, electrolyte, diaphragm, anode and packaging [6]. The diaphragm is the transport channel, which is located between the positive and negative electrodes and allows lithium ions to pass through. Figure 2 is a schematic diagram of the working principle of a LIB. Taking LiCoO2 as an example, the electrode and battery reactions during charging and discharging are given in the following equations: cathode reaction(1), anode reaction(2) and total reaction(3).
Cathode Reaction: LiCoO2 ⇌Li1-xCoO2 + xLi+ + xe- | (1) | |||||
Anode Reaction: C + xLi+ + xe- ⇌LixC | (2) | |||||
Total Reaction: C + LiCoO2 ⇌Li1-xCoO2 + LixC | (3) | |||||
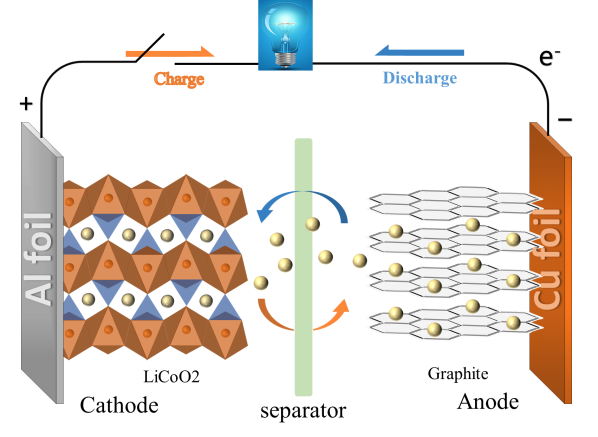
Figure 2. Working principle of a LIB.
A lithium-ion battery operates based on concentration gradients. Lithium ions are liberated from the positive electrode during the charging process and go through the separator to the negative electrode, where they are embedded. The concentration of Li simultaneously decreases at the positive electrode and rises at the negative electrode. The compensating flow of electrons occurs through the external circuit, ensuring charge balance. From the negative electrode to the positive electrode, a current is created as a result. Conversely, during discharge, one electron is lost as lithium ions flow from the negative electrode to the positive electrode through the electrolyte. As a result, the positive electrode becomes lithium-rich, and the negative electrode becomes lithium-poor. This process is accompanied by an external current flowing from the positive electrode to the negative electrode, as illustrated in Figure 2.
2.2. Types of lithium-ion batteries and their advantages and disadvantages
Table 1. Performance comparison of different types of LIBs.
LiCoO2 | LiNi1-xCoxO2 | LiNi1/3Co1/3Mn1/3O2 | LiMnO2 | LiFePO4 | |
Specific capacity mAh/g | 140 | 170 | 150 | 110 | 115 |
Cycling Performance | >500 | >500 | >800 | >300 | >2000 |
Material costs | High | Relatively high | Relatively low | Low | Low |
Safety | General | Relatively poor | Good | Good | Excellent |
Application | Small-sized batteries | Small-sized batteries | Small-sized/Traction batteries | Traction batteries | Traction batteries |
Compared to other battery types, LIBs exhibit several advantages, as depicted in Table 1 above. They possess high energy density, with the ability to achieve over 2,000 cycles for LFP (Lithium Iron Phosphate). Additionally, they offer faster charging speeds and other benefits. However, LIBs also have several drawbacks. Safety concerns arise when the current density increases, as it promotes the rapid growth of dendrite lithium, which can puncture the battery's internal structure and lead to spontaneous combustion [10]. Moreover, the limited availability of raw materials results in higher costs, as Cu foils are required for the collector, further increasing manufacturing expenses. The battery's performance is significantly affected by external environmental conditions, making it prone to malfunction in low temperatures. Lastly, the mining process for LIB materials has a severe environmental impact.
3. Sodium-ion battery
3.1. Types of lithium-ion batteries and their advantages and disadvantage
The working principle of SIBs is similar to that of LIBs, as shown in Figure 3, and SIBs are also mainly composed of a cathode, an anode, an electrolyte, and a separator [11]. The positive electrode is generally used to embed sodium-type compounds with higher potential, such as layered transition metal oxides [2], polyanionic compounds [2] and Prussian blue analogs [9]. Due to the large radius of sodium ions, anode materials with large interlayer spacing, such as hard carbon and alloy oxides, are generally chosen to ensure a high operating voltage for the full battery. Taking NaMnO2 as an example, the electrode and battery reactions during charging and discharging are given in the following equations: cathode reaction(4), anode reaction(5) and total reaction(6).
Cathode Reaction: NaMnO2 ⇌Na1-xMnO2 + xNa+ + xe- | (4) | |||||
Anode Reaction: C + xNa+ + xe- ⇌NaxC | (5) | |||||
Total Reaction: C + NaMnO2 ⇌Na1-xMnO2 +NaxC | (6) | |||||
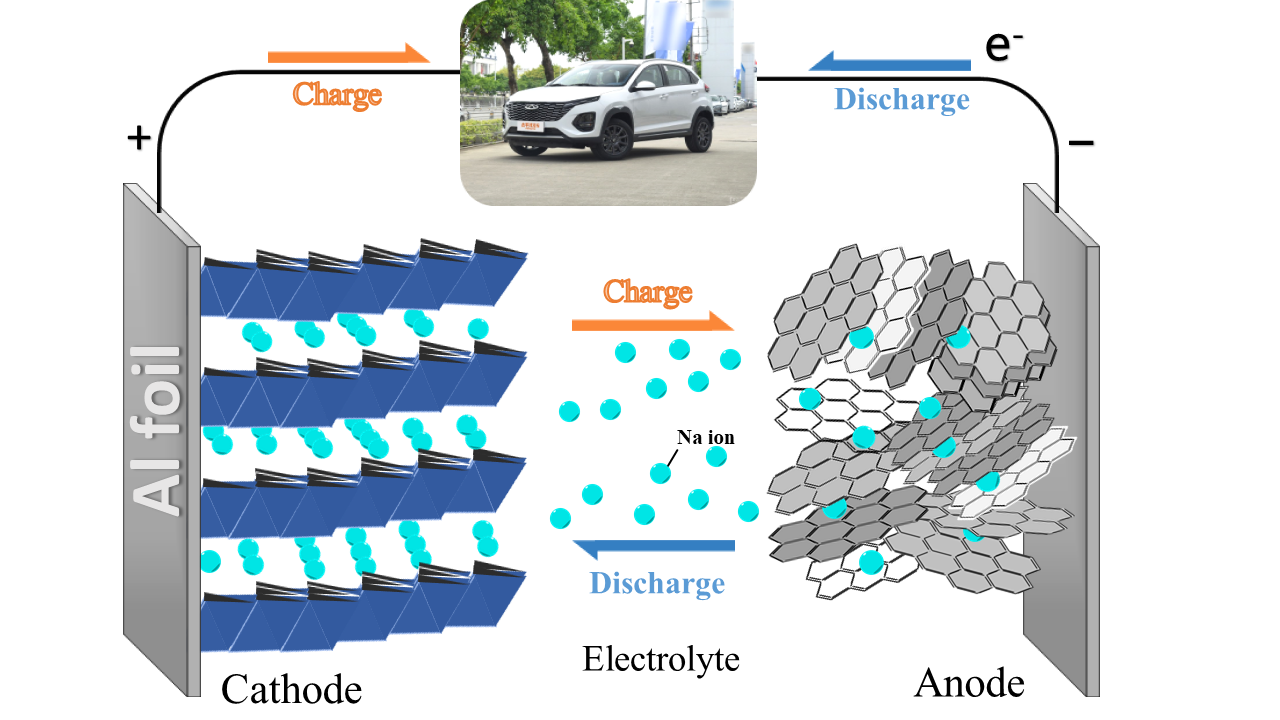
Figure 3. Working principle of a SIB.
3.2. Current progress in the industrialization of SIBs
CATL, a leading player in the new energy sector, recently organized a SIB conference. The first-generation SIBs, focusing on Prussian white material, exhibit impressive characteristics. The energy density of a single cell can reach up to 160 Wh/kg, enabling a charging time of 15 minutes at room temperature with a power output of up to 80%. Even in extremely low temperatures of -20 °C, the discharge retention remains above 90%. Moreover, the system integration efficiency can exceed 80% [4]. Other companies, such as HiNa Battery and Natriumenergy, also take great significant attention to the development of SIBs, as depicted in Table 2. HiNa Battery utilizes metal oxide as the cathode material, achieving a battery energy density of 145 Wh/kg and enabling fast charging within 6-12 minutes. It can operate within a wide temperature range of -40 to 80 ℃. Apart from CATL, there are other well-known enterprises, such as Faradion and Natron Energy, as shown in Table 2. Although Natron Energy has an energy density of 50 Wh/L, its Prussian blue aqueous electrolyte system allows for a cycle life of over 10,000 times. Additionally, companies like Sodium of the Sea of Science and Technology focus solely on raw materials, reducing battery costs by approximately 30%. Natriumenergy adopts sodium ferrate ternary cathode material, significantly increasing the battery's cycle life from 2,500 times to 10,000 times.
Table 2. Progress in the industrialization of SIBs.
Enterprise | Nation | Battery System | Performance Data | Route Advantages | Route Shortcomings |
Faradion | UK | Layered oxide/Hard carbon organic electrolyte system, 10Ah soft pack battery | Energy density 140Wh/kg, 80% DOD Cycle number 1000 times | Compatible with current lithium-ion battery manufacturing techniques | The organic system poses a safety concern, and the economic advantage is not immediately apparent. |
Natron Energy | US | Prussian Blue Aqueous Electrolyte System | Energy density 50Wh/L, 2C cycle number 10000 times | High safety, good high rate performance, and extended cycle life aqueous electrolyte system | Low energy density and complex production process |
Table 2. (continued). | |||||
Naiades | France | Sodium fluorophosphate/Hard carbon organic electrolyte system, 1Ah18650 type battery | Energy density 90Wh/kg, 1C cycle number 4000 times | Long cycle life, compatible with existing lithium-ion battery production processes | Low energy density of the system, electrode material design vanadium and fluorine elements, higher cost, greater toxicity, there are safety risks |
Natrium Energy | China | Layered oxide/Hard carbon organic electrolyte system, soft pack battery | Energy density 120Wh/kg, cycle number 2500 times | Compatible with current lithium-ion battery manufacturing techniques | The organic system poses a safety concern, and the economic advantage is not immediately apparent. |
HiNa Battery | China | Layered Oxide/Amorphous Carbon Organic Electrolyte System, Soft Pack Batteries | Energy density 135Wh/kg, cycle number 2000 times | Compatible with current lithium-ion battery manufacturing techniques | The organic system poses a safety concern, and the economic advantage is not immediately apparent. |
3.3. Advantages and disadvantages of sodium-ion batteries
SIBs offer several advantages over traditional batteries. They have a higher energy density, reaching up to 120 Wh/kg, surpassing lead-acid batteries (40 Wh/kg) and nickel-metal hydride batteries (60 Wh/kg) [4]. The wider availability of raw materials and lower costs are notable advantages. SIBs utilize Al foils for both cathode and anode collectors, which are less expensive compared to the Cu foils used in LIBs [12]. Additionally, SIBs exhibit faster charging times, longer cycle life, and enhanced safety.
SIBs, however, also have some disadvantages. They have a lower energy density compared to lithium-ion batteries, necessitating larger size and weight. The manufacturing cost is higher due to the requirement for high-quality sodium salt and other raw materials.
4. Comparison between SIBs and LIBs
4.1. Comparison of the two battery systems
SIBs and LIBs have the same structure and different materials in the battery system, as shown in Table 3. The common cathode materials for LIBs in the market are LFP, NCM, LCM, and there are three main cathode materials for SIBs studied so far, which are Transition metal oxides, Polyanionic compounds and Prussian blue analogs. Because of the small radius of lithium ions, which can be embedded between graphite layers, and the low price and good corrosion resistance, graphite material has become the mainstream anode material in the market. However, the radius of sodium ions is large, the three types of anode materials for SIBs are insertion-type, conversion-type, and alloying-type materials. Conversion type, and alloying type materials in the reaction process, sodium ions will react with host materials, the volume of the main body of the material changes drastically, resulting in the aggregation of the active material and crushing, leading to the rapid decay of the volume of the active material. Therefore, insertion-type materials are mainly used in the market at present, such as hard carbon, soft carbon, and expanded graphite. In terms of current collectors, Al foil is used for both cathode and anode electrode collectors of SIBs, which also reduces the cost of batteries to a larger extent.
Table 3. Comparison between LIBs and SIBs in battery systems.
LIBs | SIBs | |
Cathode | LFP, NCM, LCM | Transition metal oxides, Polyanionic compounds, Prussian blue analogs |
Anode | Graphite | Insertion-type, Conversion-type, Alloying-type materials |
Separator | PP, PE and other polymer films | |
Electrolyte | Lithium hexafluorophosphate | Sodium hexafluorophosphate |
Cathode current collector | Al foil | Al foil |
Anode current collector | Cu foil | Al foil |
4.2. Advantages of sodium-ion batteries
The first-generation SIB made available by CATL provides a number of benefits over lithium iron phosphate batteries, as seen in Figure 4. Despite having a lower energy density than lithium iron phosphate batteries, they are more safe, operate better in low temperatures, have faster charging speeds, and are more efficient at integrating into systems. It also provides a longer cycle life.
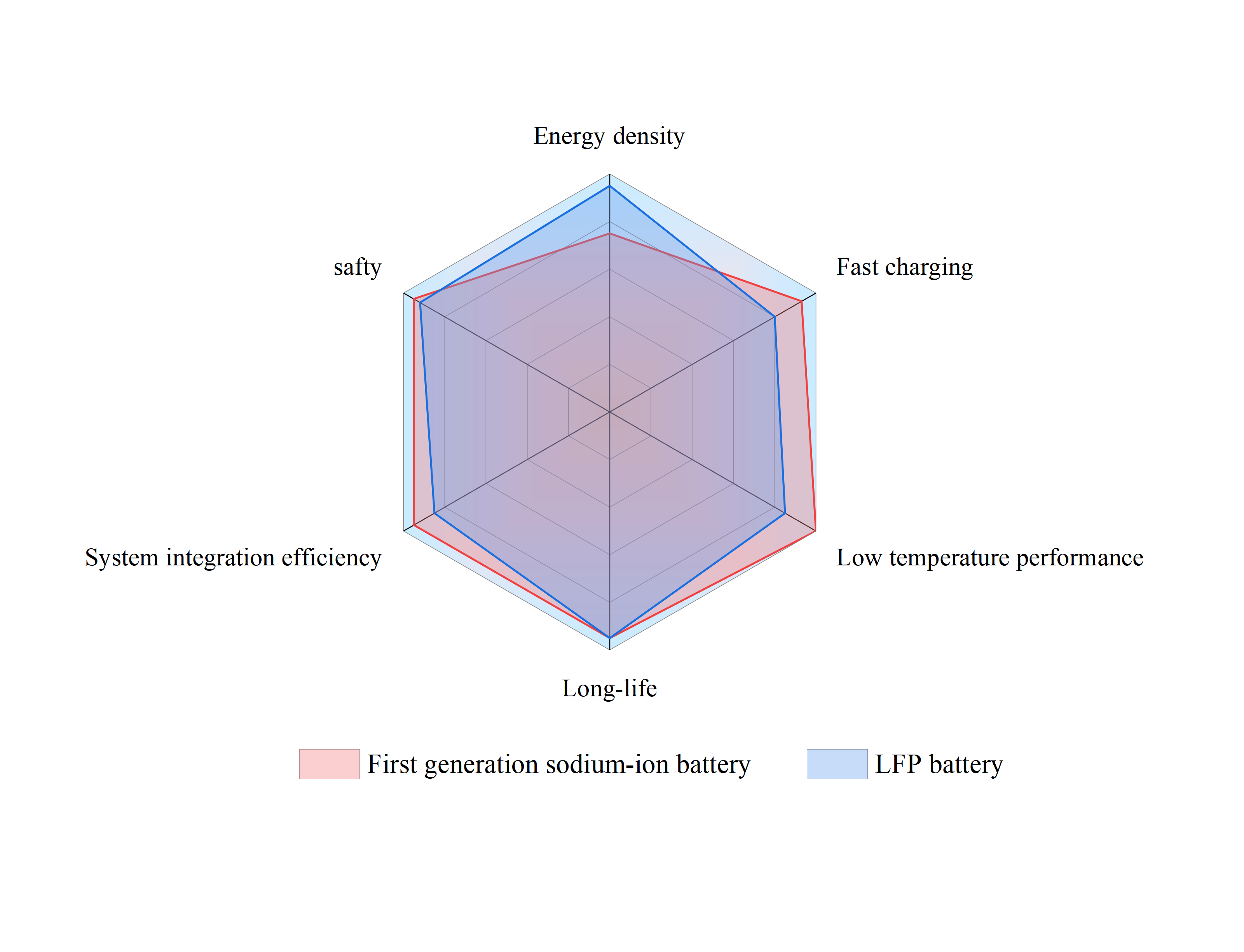
Figure 4. Performance comparison of SIBs and LIBs.
SIBs, therefore, have the potential to become one of the next-generation low-cost and high-performance energy storage battery technologies. Led by companies such as CATL, HiNa Battery, and Natrium Energy, SIBs are gradually entering the practical stage. In the coming years, the market size of SIBs will continue to expand, and their applications in electric vehicles and energy storage systems will increase [13]. With the continuous maturity and improvement of sodium-ion battery technology, it will replace a portion of the market share currently held by LIBs, becoming an important energy storage technology and making a greater contribution to clean energy and sustainable development.
5. Conclusion
This study has investigated the working principles of commonly used SIBs and LIBs available on the market. The advantages and disadvantages of SIBs and LIBs in energy storage applications have been compared. Both two types of batteries have their own advantages. LIBs exhibit low cost, abundant resources, and faster charge-discharge rates, while LIBs have higher energy density and more stable cycle life. Therefore, the specific application scenarios and requirements will also influence the choice of battery. SIBs are more suitable for energy storage applications such as solar and wind energy storage and they are expected to achieve commercialization in these fields in the future. Most of the research on SIBs, however, is still in the laboratory stage, and improvements are needed in electrode materials, electrolytes, and manufacturing technologies. Future research should focus on improving the energy density, power density, cycle stability, and lifespan of SIBs while addressing other performance aspects.
References
[1]. A G Hu, Journal of Beijing University of Technology (Social Sciences Edition), 21(3), 1-15 (2021).
[2]. X M Lin, X T Yang, H N Chen, et al. Journal of Energy Chemistry, 76, 146-164 (2023).
[3]. Q C Wang, & Y N Zhou, Design of new sodium-ion battery cathode materials and their sodium storage mechanisms. Abstract Handbook of the Third Youth Forum of the Chinese Chemical Society (2020).
[4]. T Liu, Automotive Horizon, 2023(3), 82-86 (2023).
[5]. Y T Li, Chemistry and Bioengineering, 39(9), 7-10 (2022).
[6]. H Yin, Research on the Negative Electrode Materials of Lithium-ion and Sodium-ion Batteries Based on Bismuth and Its Oxides (Doctoral dissertation). Huazhong University of Science and Technology (2017).
[7]. Z H Song, X G Jian, & F Y Hu, Acta Polymerica Sinica, 1-14 (2023).
[8]. Z Wang, Synthesis and Electrochemical Performance Study of Prussian Blue Analogues as Cathode Materials for Sodium-ion Batteries (Master's thesis). Hubei University of Technology (2022).
[9]. X Wang, Preparation and Electrochemical Performance Study of High-Performance Prussian Blue Analogues as Cathode Materials for Sodium-ion Batteries (Master's thesis). Zhejiang University (2020).
[10]. Z Ning, G Li, D L R Melvin, et al. Nature, 618(7964), 287-293(2023).
[11]. N Yabuuchi, K Kubota, M Dahbi, et al. Chemical Reviews, 114(23), 11636-11682 (2014).
[12]. K Zhang, & Y L Xu, Energy Storage Science and Technology, 12(1), 86-110. (2023)
[13]. A Rudola, R Sayers, C J Wright, et al. Nature Energy, 8(3), 215-218(2023).
Cite this article
Hua,Z. (2023). Comparative study of commercialized sodium-ion batteries and lithium-ion batteries. Applied and Computational Engineering,26,233-239.
Data availability
The datasets used and/or analyzed during the current study will be available from the authors upon reasonable request.
Disclaimer/Publisher's Note
The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of EWA Publishing and/or the editor(s). EWA Publishing and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content.
About volume
Volume title: Proceedings of the 2023 International Conference on Functional Materials and Civil Engineering
© 2024 by the author(s). Licensee EWA Publishing, Oxford, UK. This article is an open access article distributed under the terms and
conditions of the Creative Commons Attribution (CC BY) license. Authors who
publish this series agree to the following terms:
1. Authors retain copyright and grant the series right of first publication with the work simultaneously licensed under a Creative Commons
Attribution License that allows others to share the work with an acknowledgment of the work's authorship and initial publication in this
series.
2. Authors are able to enter into separate, additional contractual arrangements for the non-exclusive distribution of the series's published
version of the work (e.g., post it to an institutional repository or publish it in a book), with an acknowledgment of its initial
publication in this series.
3. Authors are permitted and encouraged to post their work online (e.g., in institutional repositories or on their website) prior to and
during the submission process, as it can lead to productive exchanges, as well as earlier and greater citation of published work (See
Open access policy for details).
References
[1]. A G Hu, Journal of Beijing University of Technology (Social Sciences Edition), 21(3), 1-15 (2021).
[2]. X M Lin, X T Yang, H N Chen, et al. Journal of Energy Chemistry, 76, 146-164 (2023).
[3]. Q C Wang, & Y N Zhou, Design of new sodium-ion battery cathode materials and their sodium storage mechanisms. Abstract Handbook of the Third Youth Forum of the Chinese Chemical Society (2020).
[4]. T Liu, Automotive Horizon, 2023(3), 82-86 (2023).
[5]. Y T Li, Chemistry and Bioengineering, 39(9), 7-10 (2022).
[6]. H Yin, Research on the Negative Electrode Materials of Lithium-ion and Sodium-ion Batteries Based on Bismuth and Its Oxides (Doctoral dissertation). Huazhong University of Science and Technology (2017).
[7]. Z H Song, X G Jian, & F Y Hu, Acta Polymerica Sinica, 1-14 (2023).
[8]. Z Wang, Synthesis and Electrochemical Performance Study of Prussian Blue Analogues as Cathode Materials for Sodium-ion Batteries (Master's thesis). Hubei University of Technology (2022).
[9]. X Wang, Preparation and Electrochemical Performance Study of High-Performance Prussian Blue Analogues as Cathode Materials for Sodium-ion Batteries (Master's thesis). Zhejiang University (2020).
[10]. Z Ning, G Li, D L R Melvin, et al. Nature, 618(7964), 287-293(2023).
[11]. N Yabuuchi, K Kubota, M Dahbi, et al. Chemical Reviews, 114(23), 11636-11682 (2014).
[12]. K Zhang, & Y L Xu, Energy Storage Science and Technology, 12(1), 86-110. (2023)
[13]. A Rudola, R Sayers, C J Wright, et al. Nature Energy, 8(3), 215-218(2023).









