1. Introduction
Adolescence is an important transition period characterized by physical and psychological changes, such as growth spurts, brain development, and sexual maturation. However, it is also a time when adolescents engage in risky behaviors. In this review, the genetic effects on adolescent behaviors was examined by focusing on the literature related to alcohol use and misuse. Then, this review will summarize the studies on how geographic factors affect the behaviors of young people and how human behaviors change the geographic environment.
2. Genetic influences on human behavior
2.1. Basic methodology of genetic epidemiology
Several different methods are available to study genetic influences at present. Among them twin studies were primarily utilized. Twin studies are a prominent method for studying genetic influences on behavior [1] This choice is particularly relevant in the current research context due to the relatively low birth rate, making twin recruitment easier. The natural occurrence of twins (around 3 per 100 births) facilitates their use in research [2].
Twins can be readily identified by registries of population-based [3] or record-based resources [4] [5]. The ease of access makes twin studies a convenient design for exploring genetic influences on behaviors. Core principle of twin studies concludes comparing the similarity of different twin types with varying degrees of genetic relatedness [6]. Monozygotic twins share 100% of their genetic material and almost identical environment, while dizygotic twins share 50% of their genes and a common environment of family. By comparing the similarities between dizygotic twins and Monozygotic twins, researchers can infer the relative importance of genes and environment. Greater similarity in MZ twins compared to DZ twins suggests a genetic influence. Similar levels of similarity between MZ and DZ twins imply shared environmental influences (e.g., family dynamics, schools, neighborhoods). Imperfect similarity even in MZ twins indicates a role for unique environmental experiences.
Twin studies have been widely applied to investigate various human behaviors, including:
• Psychopathology [7] [8]
• Personality [9] [10]
• Cognitive abilities [11] [12]
• Even seemingly less intuitive behaviors like divorce [13]
• Voting behavior [14] [15]
• Well-being and life satisfaction [16] [17]
This widespread applicability highlights the notion often referred to as "the first law of behavior genetics": all human behavioral traits likely have some degree of heritability, estimated to be around 50% [18].
However, these static heritability estimates don't capture the dynamic interplay between genes and environment throughout life. While genes influence virtually all aspects of human behavior, understanding how these influences operate is crucial. The dynamic changes that occur during adolescence make it a particularly important period for studying genetic effects. By focusing on adolescent behavior, this review can gain valuable insights into the fundamental principles of how genetic influences unfold. Figure 1 shows the changing involvement of genetic and environmental influences across adolescence in the Finnish twins : genetic influences were increasingly important, while environmental factors became less important [19] [20]
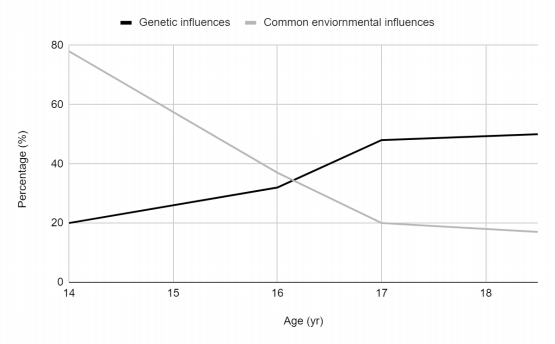
Figure 1. Percentage of Genetic Influences versus environmental influences across adolescent behaviors
2.2. Genetic influences change in importance across adolescence
Drinking alcohol is a common risk behavior among adolescents and a developmental phenomenon. Cross-sectional and longitudinal studies of alcohol use reveal age-related patterns. Longitudinal studies refer to long-term research which refer to the study of the same object over a certain period of time, while cross-sectional research refers to the study of different objects at a certain point in time. Although some children start drinking at an earlier age, alcohol use usually begins in adolescence [21]. Between the ages of 12 and 21, the proportion of alcohol use and heavy intermittent drinking increased significantly.
National surveys by SAMHSA [22] [23] show a significant increase in alcohol use among teenagers, with levels rising steadily throughout adolescence and leveling off around age 21. This includes any alcohol use, binge drinking (consuming a large amount of alcohol in a short period), and heavy use. Similar trends are observed in the Monitoring the Future study, showing a substantial rise in binge drinking prevalence from 8th to 12th grade. The frequency of binge drinking also increases during this period.
Notably, data from theNSDUH survey [24] reveals arise in the average number of binges drinking days for both genders, with a steeper increase for males. Adolescence is a critical time for the development of alcohol problems. National data suggests arise in the prevalence of past-year alcohol use disorder between ages 12 and 17, peaking in young adulthood (18-25 years old) [23]. Twin studies reveal a fascinating shift in the impact of genes and environment on alcohol use throughout adolescence. Data from Finnish twin studies [19] [25] showcases a dramatic change. Early adolescence sees environmental factors playing a more significant role, likely influenced by family, school, and neighborhood. As drinking becomes more regular, the influence of genes becomes increasingly important across adolescence. Similar findings emerge from the Virginia Adult Twin Study [26], showing a diminishing role for shared environment and a rise in genetic influence from ages 14 to 23.
Studies have identified specific genes (e.g., GABRA2 receptor gene, ALDH2) associating with alcohol use in young adulthood but not necessarily earlier. Interestingly, these genes linked to adult alcohol use might be connected to behavioral issues observed earlier in adolescence. Figure 2 shows Changes in the effect of parental monitoring on smoking frequency in children aged 14 [27]. As parental monitoring increases, genetic influences become less important, while common environmental influences become more important.
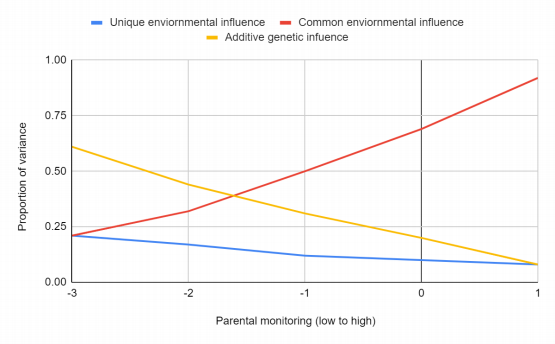
Figure 2. Proportion of variance comparing different influences (environmental vs additive genetic)
2.3. Multiple pathways of genetic risk
Twin studies reveal that susceptibility to alcohol problems isn't solely related to alcohol dependence itself. It often co-occurs with other mental health issues and externalizing behaviors like drug use, anti-social behavior, and impulsivity [28] [29] [30].
Data from the Virginia Adult Twin Study suggests that up to 65% of genes influencing alcohol dependence are also common to other disorders [28]. This "shared genetic susceptibility" likely involves reward processing and behavioral control. The remaining 35% may be specific to alcohol dependence, potentially affecting alcohol metabolism through genes like ADH andALDH2 [31] [32] [33].
Interestingly, longitudinal twin studies show a fascinating shift in the prominence of different genetic effects throughout adolescence:
2.3.1. Early Puberty
Non-specific genetic factors related to externalizing behaviors (e.g., impulsivity) play a more significant role in early puberty. This aligns with the earlier maturation of brain regions linked to reward-seeking and impulsivity compared to those involved in cognitive control [34] [35].
2.3.2. Late Puberty and Adulthood
As the brain matures, substance-specific genetic factors related to individual responses to alcohol (e.g., sensitivity) become more important. Early substance use may be influenced by broader behavioral tendencies, while regular use might depend more on specific reactions to the substance.
This pattern isn't unique to alcohol. Twin studies show similar trends with marijuana and nicotine dependence [36].
This shift in genetic influence might parallel brain development. The earlier maturation of reward- seeking regions could explain why general risk-taking behavior plays a larger role initially. Additionally, the earlier cortical maturation observed in females [37] might explain why the importance of general behavioral suppression genes declines earlier in females compared to males [38] [36].
The asynchronous development of the brain, with the striatum maturing earlier than the prefrontal cortex, is linked to increased risk-taking behavior in adolescence [39]. This developmental model aligns with the observed peak in the importance of general behavioral suppression genes during this period [40].
2.3.3. Environment moderates the importance of genetic influences
Adolescence is a wealthy field for the study of gene-environmental interaction, because in this stage of development, there have been many changes in the relevant environmental factors. This has become an increasingly interesting research field. Progress has been made in the statistical modeling of these more complex interactions [41] [42]. Twin studies show that in the presence of friends who use drugs [43] and in environments with low parental monitoring levels, the genetic impact on adolescent drug use is greater [27]. Figure 2 shows a huge change in the relative importance of genetic effects in different environments using the data of the Finnish project of twin: at the extremely low end of parental monitoring, the genetic effect plays the greatest role in affecting adolescent smoking, while in families with a high degree of parental monitoring, the genetic effect is almost absent. The common factors of environmental are the most important impact [27]. Similar effects have been proved for more generalized behaviors: illegal peers [44], high negative feelings by parents, low parental warms [45], and high patrilineal punitive discipline [46] lead to the higher impact of heredity on antisocial behavior, and more general environmental adversity [47]. The influence at the social area or neighborhood level has also been shown to mitigate the importance of genetic influence on the use of drug. In urban environments, communities characterized by a large number of immigrants and communities with a higher proportion of older adolescents/young people, the impact of heredity on alcohol use in late adolescence and early adolescent behavior is enhanced [48] [49] [19]. These moderate effects may reflect alcohol supply, a series of possible different examples, neighborhood stability, and differences at the community level in different regions. The environment that regulates the genetic impact of adolescence seems to largely reflect different social controls and/or opportunities, leading to different expressions of personal preferences [50]. In addition, the importance of different environments as regulatory factors for genetic effects may vary according to the stage of development. There are some signs in the data of Finnish data of twin, where parental monitoring showed significant moderating effects on substance use beginning in early adolescence (14 years of age), and peer substance use moderating effects until late adolescence (age 17 [43]).Similarly, the socioregional impact of easing alcohol use in early adulthood shows no evidence of moderate alcohol use in the early stages of adolescents [48]. Therefore, the study of the impact of heredity for adolescent behavior shows that genetic factors can only be understood in the context of the environment, and these interactions may also change during development.
2.4. Blurring the Lines: Genes, Environment, and Adolescent Behavior
It's argued that separating genetic and environmental influences is misleading [51]. Many environmental factors, like family environment, peer relationships, and stressful life events, show evidence of being partially influenced by genes themselves. This "heritability of environment" is estimated to be between 15-35% [51]. As individuals, particularly adolescents with greater autonomy, have more control over shaping their environment, this influence becomes more pronounced.
Twin studies illustrate how genes influence an adolescent's choice of peers [52] [8]. The impact of genetic factors on peer group deviation increases throughout adolescence. While only explaining 30% of the variance between ages 8 and 11, this influence rises to 50% by age 15 [53]. This suggests that genetic predispositions may interact with increasing autonomy to shape an adolescent's social environment, potentially influencing alcohol use.
Multiple methods are used to understand the genetic architecture of alcohol use, including:
• Linkage analysis: Identifying genomic regions shared within families affected by alcohol use [54] [55].
• Candidate gene association studies: Testing specific genes potentially linked to alcohol use or related behaviors [56].
• Genome-wide association studies (GWAS): Systematically scanning the entire genome for associations with a phenotype.
Among these methods:
• Linkage analysis allows for unbiased genome-wide exploration but struggles with complex gene interactions and environmental influences [57]. However, recent advancements in whole genome sequencing analysis are showing promise [58].
• Candidate gene association studies are statistically robust but require a priori knowledge of potentially relevant genes [59]. These studies can be conducted within families (testing associations) or across families.
• GWAS combines the unbiased nature of linkage analysis with the statistical power of association studies [60]. It leverages advancements in single nucleotide polymorphisms (SNPs) to scan the entire genome for potential associations ([61] The International HapMap Project; The 1000 Genomes Project). However, most SNPs have no clear impact, highlighting the importance of genetic diversity.
3. Environmental influences on the human body, cognition, and activity
The profound impact of the natural environment on human health and well-being is a subject of growing concern across disciplines. As this review navigates the modern urban landscape, it becomes increasingly important to understand the complex relationship between nature and our physical, cognitive, and behavioral aspects. This article will delve into the impact of the natural environment on the human body, cognition, and activity, supported by scientific research and illustrated by real- world examples.
3.1. Sunlight and vitamin D synthesis
Sunlight is an essential component of the natural environment and is the catalyst for vitamin D synthesis in the human body which plays a key role in bone health, immune function, and overall health. People living in areas with limited sunlight, especially during certain seasons, are more likely to be deficient in vitamin D. For example, residents of northern European countries experience less sunlight in winter, leading to higher rates of vitamin D deficiency.
3.2. Green space and stress relief
Green spaces, such as parks and forests, have been linked to reduced stress and improved mental health. Incorporating greenery into urban environments can have a significant impact on reducing stress. Cities like Singapore, for example, integrate lush green spaces such as Gardens by the Bay to provide residents with a peaceful environment amid the city's hustle and bustle.
3.3. Cognitive recovery
The natural environment has a profound effect on cognitive function. Spending time in nature improves focus and creativity while reducing mental fatigue. Forest environments, in particular, have shown benefits for cognitive recovery. The Japanese Shinrin-yoku is a testament to conscious immersion in a natural environment to improve cognitive function and emotional health.
3.4. Urban green space and sports activities
Access to green space in an urban environment has a direct impact on physical activity levels. It was found that individuals who live in neighborhoods with more green space are more likely to participate in physical activity on a regular basis. New York City's High Line, a converted elevated rail Line, is a model of how urban green Spaces can be transformed into recreational corridors that encourage physical activity and community interaction.
3.5. Waterfront and Social interaction
Proximity to natural bodies of water, such as rivers and lakes, has been linked to increased social interaction and community cohesion. The Blue Health project explored the health benefits of "blue spaces" and found that living near water was associated with higher levels of physical activity and enhanced mental health. Vancouver’s seawall, ascenic passage along the waterfront, exemplifies how urban planning uses natural elements to promote physical activity and social connections.
3.6. Relative humidity
Relative humidity can be expressed as a ratio. The degree to which the air is near saturation can be reported as a percentage: if the relative humidity is 100%, then the air is saturated. However, if the relative humidity is 50%, the air contains half of the water vapor needed to saturate it. It follows, therefore, that as the amount of water vapor in the air increases, the relative humidity increases; Conversely, if the amount of water vapor in the air decreases, the relative humidity of the air will also decrease. However, relative humidity also depends on the temperature of the air. If the water vapor content is flat and the temperature decreases, the relative humidity of the air will still increase.
3.7. Dew point
Dew point is a better humidity indicator than relative humidity because it is not a temperature- dependent percentage. The dew point is the temperature at which the air must be cooled to saturation. Below the dew point, water condenses from the air onto the surface. In the early morning, if the overnight temperature drops below the dew point, the grass surface will be covered with water. When the humidity is high, the dew point temperature is only a few degrees lower than the air temperature or equal to the air temperature. In dry places, such as deserts in the Southwest, temperatures can be 50 or 60 degrees above the dew point. In general, dew point is a more reliable indicator of humidity than relative humidity because it is not affected by changes in air temperature and doesn’t fluctuate much throughout the day.
3.8. Effects on human health and comfort
Temperature and humidity affect people's comfort and health. High humidity and heat mean there is more moisture in the air, which further carries odor molecules, causing a considerable stench around bacterial sources such as garbage in the summer. Recent studies have revealed links between humidity, public health and temperature. In temperate regions of the world, humidity and temperature directly affect the spread of influenza viruses. In temperate regions of each hemisphere, influenza activity increases in winter. The colder it is outside, the more the flu virus flourishes. The relative humidity is high in winter, but due to heating, the indoor relative humidity is relatively dry. Exposure to cold outdoor air and dry indoor air increases the spread of the flu virus. Studies have shown that aerosolized influenza viruses are more stable at lower relative humidity. At high temperatures, the half-life of the virus is shortened and it is not easy to spread.
Changes in temperature and humidity can also lead to heart risks. Cardiovascular mortality increased under low temperature and high humidity conditions. This may be due to high humidity affecting the risk of blood clots, combined with the body's various cold stress responses. When this review uses scientific articles to help you understand how humidity and temperature affect your health, you should keep one important message in mind: there is no unique relationship between temperature and humidity. However, at the same temperature, humidity changes a person's comfort level because humans are sensitive to the amount of water vapor in the atmosphere.
In cold climates, outdoor temperatures reduce the ability of water vapor to move. So although it may be snowing and the humidity is high relative to the temperature outside, once the air enters the building and is heated, its new relative humidity will be very low, making the air very dry and causing discomfort.
4. Human impacts on environment
4.1. Climate change
The burning of fossil fuels (coal, oil, natural gas) is a primary source of greenhouse gases. Deforestation and land-use changes related to fossil fuel exploration and extraction, refining, energy generation, and industrial agriculture all contribute to the global carbon cycle disruption. Livestock production and associated land use changes, including deforestation for grazing, are responsible for an estimated 18% of global greenhouse gas emissions. Increased greenhouse gas concentrations lead to potentially severe consequences, including: Changes in temperature and weather patterns (more frequent and severe droughts, floods), Sea level rise, Ecosystem destruction.
4.2. Acid Deposition (Acid Rain)
Fossil fuel combustion releases pollutants that form sulfuric and nitric acids in the atmosphere. Acid rain contaminates rivers, lakes, wetlands, and other aquatic ecosystems. It damages forests, depletes soil nutrients, releases aluminum that harms soil fertility, and hinders water absorption by plants.
4.3. Ozone Depletion and the Ozone Hole
Observed since the late 1970s, ozone depletion mainly affects the polar regions (ozone holes). The primary culprit is the use of man-made chemicals, particularly halogenated hydrocarbons used in refrigerants, solvents, propellants, and foam blowing agents (ozone-depleting substances). Concerns about increased risks of skin cancer, sunburn, cataracts, and other negative effects due to increased UV radiation exposure led to the Montreal Protocol of 1987. This international agreement banned the production of chlorofluorocarbons, halons, and other ozone-depleting substances.
4.4. Disruption of the Nitrogen Cycle
Nitrous oxide (N2O) emissions from industrial processes, vehicles, agricultural fertilization, and livestock contribute to nitrogen cycle disruption. N2O has along atmospheric lifetime (114-120 years) and a potent greenhouse effect (300 times that of CO2). Disruptions in the nitrogen cycle can lead to altered nutrient cycling and ecosystem health.
4.5. Energy Consumption and its Environmental Impact
The reliance on fossil fuels for energy generation contributes to global warming and climate change. Biodiesel, while a cleaner alternative to traditional diesel, still has environmental drawbacks. Requires energy for its production. Emits higher levels of certain pollutants compared to diesel. Coal extraction processes (drilling, blasting) release significant air pollutants. These pollutants can cause respiratory and cardiovascular illnesses.
5. Conclusion
This review underscores the intricate interplay between genetics, geography, and human behavior, particularly in the formative adolescent years. By examining the methodologies of genetic epidemiology and contrasting genetic and environmental influences on adolescent behaviors, this study illuminates the complex pathways through which genetic predispositions manifest in the context of specific geographic environments. The findings suggest that while genetic factors contribute significantly to behavioral outcomes, the geographic environment shapes these expressions, often in interactive ways. This research not only deepens our understanding of human development but also provides a foundation for designing interventions that optimize both genetic potential and environmental conditions, fostering healthier and more productive adolescent outcomes.
References
[1]. Plomin, R., DeFries,J.C., McClearn, G.E., McGuffin, P., 2001. Behavioral Genetics. Worth, London.
[2]. Hamilton, B.E., Martin, J.A., Osterman, M., Curtin, S.C., Matthews, T.J., 2015. Births: Final Data for 2014. National Center for Health Statistics, Hyattsville, MD.
[3]. Kaprio, J., Pulkkinen, L., Rose, R.J., 2002. Genetic and environmental factors in health-related behaviors: studies on Finnish twins and twin families. Twin Res. 5, 358–365.
[4]. Anderson, L.S., Beverly, W.T., Corey, L., Murrelle,L., 2002. The mid-Atlantic twin registry. Twin Res. 5, 449–455.
[5]. Meyer, J.M., Silberg, J.L., Simonoff, E., Kendler, K.S., Hewitt, J.K., 1996. The Virginia twin-family study of adolescent behavioral development: assessing sample biases in demographic correlates of psychopathology. Psychol. Med. 26, 1119–1133.
[6]. Turkheimer, E., Waldron, M., 2000. Nonshared environment: a theoretical, methodological, and quantitative review. Psychol. Bull. 126, 78–108.
[7]. Hewitt,J.K., Silberg, J.L., Rutter, M., Simonoff, E., Meyer, J.M., Maes, H.H., Pickles, A., Neale, M.C., Loeber, R., Erickson, M.T., Kendler, K.S., Health, A.C., Truett, K.R., Reynolds, C.A., Eaves, L.J., 1997. Genetics and developmental psycholpathology: 1. Pehnotypic assessment in the Virginia Twin Study of Adolescent Behavioral Development. J. Child Psychol. Psychiatry 38, 943–963.
[8]. McGue, M., Iacono, W.G., Krueger, R., 2006. The association of early adolescent problem behavior and adult psychopathology: a multivariate behavioral genetic perspective. Behav. Genet. 36, 591–602.
[9]. Littlefield, A.K., Agrawal,A., Ellingson, J.M., Kristjansson, S., Madden, P.A., Bucholz, K.K., Slutske, W.S., Heath, A.C., Sher, K.J., 2011. Does variance in drinking motives explain the genetic overlap between personality and alcohol use disorder symptoms?A twin study of young women alcoholism. Clin. Exp. Res. 35, 2242–2250.
[10]. Viken, R.J., Kaprio, J., Rose, R.J., 2007. Personality at ages 16 and 17 and drinking problems at ages 18 and 25: genetic analyses of data from FinnTwin16-25. Twin Res. Hum. Genet. 10, 25—32.
[11]. Plomin, R., DeFries,J.C., 1998. The genetics of cognitive abilities and disabilities. Sci. Am. 278, 62—69.
[12]. Trzaskowski, M., Davis, O.S., DeFries, J.C., Yang, J., Visscher, P.M., Plomin, R., 2013. DNA evidence for strong genome-wide pleiotropy of cognitive and learning abilities. Behav. Genet. 43, 267—273.
[13]. McGue,M., Lykken, D.T., 1992. Genetic influence on risk of divorce. Psychol. Sci. 3, 368—373.
[14]. Eaves, L.J., Heath, A., Martin, N., Maes, H.H., Neale, M.C., Kenderl, K.S., Kirk, K., Corey, L., 1999. Comparing the biological and cultural inheritance of personality and social attitutdes in the Virginia 30,000 study of twin and their relatives. Twin Res. 2, 62—80.
[15]. Hatemi,P.K., Smith, K., Alford, J.R., Martin, N.G., Hibbing, J.R., 2015. The genetic and envrionmental foundations of political, psychological, social, and economic behaviors: a panel study of twins and families. Twin Res. Hum. Genet. 18, 243—255.
[16]. Archontaki, D., Lewis, G.J., Bates, T.C., 2013. Genetic influence on psychological well-being: a nationally representative twin study. J. Pers. 81, 221—230.
[17]. Sadler,M.E., Miller, C.J., Christensen, K., McGue,M., 2011. Subjective wellbeing and longevity: a co-twin control study. Twin Res. Hum. Genet. 14, 249—256.
[18]. Turkheimer, E., 2000. Three laws of behavior genetics and what they mean. Curr. Dir. Psychol. Sci. 9, 160—164.
[19]. Rose, R.J., Dick, D.M., Viken, R.J., Kaprio, J., 2001a. Gene-environment interaction in patterns of adolescent drinking: regional residency moderates longitudinal influences on alcohol use. Alcohol.: Clin. Exp. Res. 25, 637— 643.
[20]. Rose, R.J., Viken, R.J., Dick, D.M., Bates, J., Pulkkinen, L., Kaprio, J., 2003. It does take a village: nonfamilial environments and children’s behavior. Psychol. Sci. 14, 273—277.
[21]. Faden, F.B., 2006. Trends in initiation of alcohol use in the United States 1975 to 2003. Clin. Exp. Res. 30, 1011— 1022.
[22]. SAMHSA, 2007. Results from the 2006 National Survey on Drug Use and Health: National Findings. Office of Applies Studies, Rockville, MD.
[23]. SAMHSA, 2014. Results from the 2013 National Survey on Drug Use and Health: Summary of National Findings. Office of Applied Statistics, Rockville, MD.
[24]. Chen, C.M., Yi, H., Faden, V.B., 2015. Trends in Underage Drinking in the United States 1991—2013. National Institute on Alcohol Abuse and Alcoholism Bethesda, MD.
[25]. Rose, R.J., Dick, D.M., Viken, R.J., Pulkkinen, L., Kaprio, J., 2001b. Drinking or abstaining at age 14: a genetic epidemiological study. Alcohol.: Clin. Exp. Res. 25, 1594—1604.
[26]. Kendler, K.S., Schmitt, E., Aggen, S.H., Prescott, C.A., 2008b. Genetic and environmental influences on alcohol, caffeine, cannabis, and nicotine use from early adolescence to middle adulthood. Arch. Gen. Psychiatry 65, 674— 682.
[27]. Dick, D.M., Viken, R., Purcell, S., Kaprio, J., Pulkkinen, L., Rose, R.J., 2007b. Parental monitoring moderates the importance of genetic and environmental influences on adolescent smoking. J. Ab
[28]. Kendler, K.S., Prescott, C., Myers, J., Neale, M.C., 2003. The structure of genetic and environmental risk factors for common psychiatric and substance use disorders in men and women. Arch. Gen. Psychiatry 60, 929—937.
[29]. Krueger,R.F., Hicks, B.M., Patrick, C.J., Carlson, S.R., Iacono, W.G., McGue,M., 2002.
[30]. Young, S.E., Stallings, M.C., Corley, R.P., Krauter,K.S., Hewitt,J.K., 2000. Genetic and environmental influences on behavioral disinhibition. Am. J. Med. Genet. 96, 684—695.
[31]. Edenberg, H.J., Xuei, X., Chen, H.J., Tian, H., Wetherill, L.F., Dick, D.M., Almasy, L., Bierut, L., Bucholz, K.K., Goate,A., Hesselbrock, V., Kuperman, S., Nurnberger,J., Porjesz, B., Rice,J., Schuckit,M., Tischfield, J., Begleiter, H., Foroud, T., 2006. Association of alcohol dehydrogenase genes with alcohol dependence: a comprehensive analysis. Hum. Mol. Genet. 15, 1539—1549.
[32]. Kuo, P.H., Kalsi, G., Prescott, C.A., Hodgkinson, C.A., Goldman, D., van den Oord, E.J., Alexander,J., Jiang, C., Sullivan, P.F., Patterson, D.G., Walsh, D., Kendler, K.S., Riley, B.P., 2008. Association of ADHandALDH genes with alcohol dependence in the Irish Affected Sib Pair Study of alcohol dependence (IASPSAD) sample. Alcohol.: Clin. Exp. Res. 32, 785—795.
[33]. Whitfield, J.B., 1997. Meta-analysis of the effects of alcohol dehydrogenase genotype on alcohol dependence and alcoholic liver disease. Alcohol Alcohol. 32, 613—619.
[34]. Casey, B.J., Galvan, A., Hare, T.A., 2005. Changes in cerebral functional organization during cognitive development. Curr. Opin. Neurobiol. 15, 239—244.
[35]. Clark,D.B., Winters, K.C., 2002. Measuring risks and outcomes in substance use disorders prevention research. J. Consult. Clin. Psychol. 70, 1207—1223.
[36]. Vrieze, S.I., Hicks, B.M., Iacono, W.G., McGue, M., 2012. Decline in genetic influence on the co-occurrence of alcohol,marijuana, and nicotine dependence symptoms from age 14 to 29. Am. J. Psychiatry 169, 1073—1081.
[37]. Lenroot, R.K., Gogtay, N., Greenstein, D.K., Wells, E.M., Wallace, G.L., Clasen, L.S., Blumenthal, J., Lerch, J., Zijdenbos, A.P., Evans, A.C., Thompson, P.M., Giedd, J., 2007. Sexual dimorphism of brain developmental trajectories during childhood and adolescence. Neuroimage 36, 1065–1073.
[38]. Meyers, J.L., Salvatore,J.E., Vuoksimaa, E., Korhonen, T., Pulkkinen, L., Rose, R.J., Kaprio, J., Dick, D.M., 2014. Genetic influences on alcohol use behaviors have diverging developmental trajectories: a prospective study among male and female twins. Alcohol.: Clin. Exp. Res. 38, 2869–2877.
[39]. Steinberg, L., 2004. Risk taking in adolescence: what changes, and why. Ann. N. Y. Acad. Sci. 1021, 51–58.
[40]. Steinberg, L., Graham, S., O’Brien, L., Woolard, J., Cauffman, E., Banich, M., 2009. Age differences in future orientation and delay discounting. Child Dev. 80, 28–44.
[41]. Purcell, S., 2002. Variance components models for gene-environment interaction in twin analysis. Twin Res. 5, 554–571.
[42]. van der Sluis, S., Posthuma, D., Dolan, C.V., 2012. A note of false positives and power in GxE modelling of twin data. Behav. Genet. 42, 170–186.
[43]. Dick, D.M., Pagan, J.L., Viken, R., Purcell,S., Kaprio, J., Pulkkinen, L., Rose,R.J., 2007a. Changing environmental influences on substance use across development. Twin Res. Hum. Genet. 10, 315–326.
[44]. Button, T.M., Rhee, S.H., Hewitt,J.K., Young, S.E., Corley,R.P., Stallings, M.C., 2007.The role of conduct disorder in explaining the comorbidity between alcohol and illicit drug dependence in adolescence. Drug Alcohol Depend. 87, 46–53.
[45]. Feinberg, M.E., Button, T.M., Neiderhiser, J.M., Reiss, D., Hetherington, E.M., 2007.
[46]. Button, T.M., Lau, J.Y., Maughan, B., Eley, T.C., 2008. Parental punitive discipline, negative life events and gene-environment interplay in the development of externalizing behavior. Psychol. Med. 38, 29–39.
[47]. Hicks, B.M., South, S., DiRago, A.C., Iacono, W.G., McGue, M., 2009. Environmental adversity and increasing genetic risk for externalizing disorders. Arch. Gen. Psychiatry 66, 640–648.
[48]. Dick, D.M., Bernard, M., Aliev,F., Viken, R., Pulkkinen, L., Kaprio, J., Rose,R.J., 2009a. The role of socioregional factors in moderating genetic influences on early adolescent behavior problems and alcohol use. Alcohol.: Clin. Exp. Res. 33, 1739–1748.
[49]. Dick, D.M., Rose, R.J., Viken, R.J., Kaprio, J., Koskenvuo, M., 2001. Exploring gene-environment interactions: socioregional moderation of alcohol use. J. Abnorm. Psychol. 110, 625–632.
[50]. Shanahan, M.J., Hofer, S.M., 2005. Social context in gene-environment interactions: retrospect and prospect. J. Gerontol. Ser. B: Psycholo. Sci. Social Sci. 60 (Spec. No. 1), 65–76.
[51]. Kendler,K.S., Baker, J.H., 2007. Genetic influences on measures of the environment: a systematic review. Psychol. Med. 37, 615–626.
[52]. Kendler, K.S., Jacobson, K., Myers, J.M., Eaves, L.J., 2008a. A genetically informative developmental study of the relationship between conduct disorder and peer deviance in males. Psychol. Med. 38, 1001–1011.
[53]. Kendler,K.S., Jacobson, K.C., Gardner, C., Gillespie,N.A., Aggen, S.H., Prescott, C., 2007. Creating a social world: a developmental twin study of peer group deviance. Arch. Gen. Psychiatry 64, 958–965.
[54]. Dick, D.M., Agrawal, A., Keller, M.C., Adkins, A., Aliev, F., Monroe, S., Hewitt, J.K., Kendler, K.S., Sher, K.J., 2015. Candidate gene-environment interaction research: reflections and recommendations. Perspect. Psychol. Sci. 10, 37–59.
[55]. Saccone, N.L., Downey, J.T.J., Meyer, D.J., Neuman, R.J., Rice, J.P., 1999. Mapping genotype to phenotype for linkage analysis. Genet. Epidemiol. 17 (Suppl. 1), S703–S708.
[56]. Wang, S., Huang, S., Liu, N., Chen, L., Oh, C., Zhao, H., 2005. Whole-genome linkage analysis in mapping alcoholism genes using single-nucleotide polymorphisms and microsatellites. BMC Genet. 6 (Suppl. 1), S28.
[57]. Hirschhorn, J.N., Daly, M., 2005. Genome-wide association studies for common diseases and complex traits. Nat. Rev. – Genet. 6, 95–108.
[58]. Ott,J., Wang, J., Leal,S., 2015. Genetic linkage analysis in the age of whole-genome sequencing. Nat. Rev. – Genet. 16, 275–284.
[59]. Risch, N., Merikangas, K., 1996. The future of genetic studies of complex human diseases. Science 273, 1516–1517.
[60]. McCarthy, M.I., Abecasis, G.R., Cardon, L.R., Goldstein, D.B., Little, J., Ioannidis, J.P., Hirschhorn, J.N., 2008. Genome-wide association studies for complex traits: consensus, uncertainty and challenges. Nat. Rev. – Genet. 9, 356–369.
[61]. Barrett,J.C., Fry,B., Maller, J., Daly,M.J., 2005. Haploview: analysis and visualization of LD and haplotype maps. Bioinformatics 21, 263–265.
Cite this article
Chen,S. (2024). Impacts of genetics and geoscience on the behaviors of young people and how human behaviors change the geographic environment. Theoretical and Natural Science,63,27-37.
Data availability
The datasets used and/or analyzed during the current study will be available from the authors upon reasonable request.
Disclaimer/Publisher's Note
The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of EWA Publishing and/or the editor(s). EWA Publishing and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content.
About volume
Volume title: Proceedings of the 4th International Conference on Biological Engineering and Medical Science
© 2024 by the author(s). Licensee EWA Publishing, Oxford, UK. This article is an open access article distributed under the terms and
conditions of the Creative Commons Attribution (CC BY) license. Authors who
publish this series agree to the following terms:
1. Authors retain copyright and grant the series right of first publication with the work simultaneously licensed under a Creative Commons
Attribution License that allows others to share the work with an acknowledgment of the work's authorship and initial publication in this
series.
2. Authors are able to enter into separate, additional contractual arrangements for the non-exclusive distribution of the series's published
version of the work (e.g., post it to an institutional repository or publish it in a book), with an acknowledgment of its initial
publication in this series.
3. Authors are permitted and encouraged to post their work online (e.g., in institutional repositories or on their website) prior to and
during the submission process, as it can lead to productive exchanges, as well as earlier and greater citation of published work (See
Open access policy for details).
References
[1]. Plomin, R., DeFries,J.C., McClearn, G.E., McGuffin, P., 2001. Behavioral Genetics. Worth, London.
[2]. Hamilton, B.E., Martin, J.A., Osterman, M., Curtin, S.C., Matthews, T.J., 2015. Births: Final Data for 2014. National Center for Health Statistics, Hyattsville, MD.
[3]. Kaprio, J., Pulkkinen, L., Rose, R.J., 2002. Genetic and environmental factors in health-related behaviors: studies on Finnish twins and twin families. Twin Res. 5, 358–365.
[4]. Anderson, L.S., Beverly, W.T., Corey, L., Murrelle,L., 2002. The mid-Atlantic twin registry. Twin Res. 5, 449–455.
[5]. Meyer, J.M., Silberg, J.L., Simonoff, E., Kendler, K.S., Hewitt, J.K., 1996. The Virginia twin-family study of adolescent behavioral development: assessing sample biases in demographic correlates of psychopathology. Psychol. Med. 26, 1119–1133.
[6]. Turkheimer, E., Waldron, M., 2000. Nonshared environment: a theoretical, methodological, and quantitative review. Psychol. Bull. 126, 78–108.
[7]. Hewitt,J.K., Silberg, J.L., Rutter, M., Simonoff, E., Meyer, J.M., Maes, H.H., Pickles, A., Neale, M.C., Loeber, R., Erickson, M.T., Kendler, K.S., Health, A.C., Truett, K.R., Reynolds, C.A., Eaves, L.J., 1997. Genetics and developmental psycholpathology: 1. Pehnotypic assessment in the Virginia Twin Study of Adolescent Behavioral Development. J. Child Psychol. Psychiatry 38, 943–963.
[8]. McGue, M., Iacono, W.G., Krueger, R., 2006. The association of early adolescent problem behavior and adult psychopathology: a multivariate behavioral genetic perspective. Behav. Genet. 36, 591–602.
[9]. Littlefield, A.K., Agrawal,A., Ellingson, J.M., Kristjansson, S., Madden, P.A., Bucholz, K.K., Slutske, W.S., Heath, A.C., Sher, K.J., 2011. Does variance in drinking motives explain the genetic overlap between personality and alcohol use disorder symptoms?A twin study of young women alcoholism. Clin. Exp. Res. 35, 2242–2250.
[10]. Viken, R.J., Kaprio, J., Rose, R.J., 2007. Personality at ages 16 and 17 and drinking problems at ages 18 and 25: genetic analyses of data from FinnTwin16-25. Twin Res. Hum. Genet. 10, 25—32.
[11]. Plomin, R., DeFries,J.C., 1998. The genetics of cognitive abilities and disabilities. Sci. Am. 278, 62—69.
[12]. Trzaskowski, M., Davis, O.S., DeFries, J.C., Yang, J., Visscher, P.M., Plomin, R., 2013. DNA evidence for strong genome-wide pleiotropy of cognitive and learning abilities. Behav. Genet. 43, 267—273.
[13]. McGue,M., Lykken, D.T., 1992. Genetic influence on risk of divorce. Psychol. Sci. 3, 368—373.
[14]. Eaves, L.J., Heath, A., Martin, N., Maes, H.H., Neale, M.C., Kenderl, K.S., Kirk, K., Corey, L., 1999. Comparing the biological and cultural inheritance of personality and social attitutdes in the Virginia 30,000 study of twin and their relatives. Twin Res. 2, 62—80.
[15]. Hatemi,P.K., Smith, K., Alford, J.R., Martin, N.G., Hibbing, J.R., 2015. The genetic and envrionmental foundations of political, psychological, social, and economic behaviors: a panel study of twins and families. Twin Res. Hum. Genet. 18, 243—255.
[16]. Archontaki, D., Lewis, G.J., Bates, T.C., 2013. Genetic influence on psychological well-being: a nationally representative twin study. J. Pers. 81, 221—230.
[17]. Sadler,M.E., Miller, C.J., Christensen, K., McGue,M., 2011. Subjective wellbeing and longevity: a co-twin control study. Twin Res. Hum. Genet. 14, 249—256.
[18]. Turkheimer, E., 2000. Three laws of behavior genetics and what they mean. Curr. Dir. Psychol. Sci. 9, 160—164.
[19]. Rose, R.J., Dick, D.M., Viken, R.J., Kaprio, J., 2001a. Gene-environment interaction in patterns of adolescent drinking: regional residency moderates longitudinal influences on alcohol use. Alcohol.: Clin. Exp. Res. 25, 637— 643.
[20]. Rose, R.J., Viken, R.J., Dick, D.M., Bates, J., Pulkkinen, L., Kaprio, J., 2003. It does take a village: nonfamilial environments and children’s behavior. Psychol. Sci. 14, 273—277.
[21]. Faden, F.B., 2006. Trends in initiation of alcohol use in the United States 1975 to 2003. Clin. Exp. Res. 30, 1011— 1022.
[22]. SAMHSA, 2007. Results from the 2006 National Survey on Drug Use and Health: National Findings. Office of Applies Studies, Rockville, MD.
[23]. SAMHSA, 2014. Results from the 2013 National Survey on Drug Use and Health: Summary of National Findings. Office of Applied Statistics, Rockville, MD.
[24]. Chen, C.M., Yi, H., Faden, V.B., 2015. Trends in Underage Drinking in the United States 1991—2013. National Institute on Alcohol Abuse and Alcoholism Bethesda, MD.
[25]. Rose, R.J., Dick, D.M., Viken, R.J., Pulkkinen, L., Kaprio, J., 2001b. Drinking or abstaining at age 14: a genetic epidemiological study. Alcohol.: Clin. Exp. Res. 25, 1594—1604.
[26]. Kendler, K.S., Schmitt, E., Aggen, S.H., Prescott, C.A., 2008b. Genetic and environmental influences on alcohol, caffeine, cannabis, and nicotine use from early adolescence to middle adulthood. Arch. Gen. Psychiatry 65, 674— 682.
[27]. Dick, D.M., Viken, R., Purcell, S., Kaprio, J., Pulkkinen, L., Rose, R.J., 2007b. Parental monitoring moderates the importance of genetic and environmental influences on adolescent smoking. J. Ab
[28]. Kendler, K.S., Prescott, C., Myers, J., Neale, M.C., 2003. The structure of genetic and environmental risk factors for common psychiatric and substance use disorders in men and women. Arch. Gen. Psychiatry 60, 929—937.
[29]. Krueger,R.F., Hicks, B.M., Patrick, C.J., Carlson, S.R., Iacono, W.G., McGue,M., 2002.
[30]. Young, S.E., Stallings, M.C., Corley, R.P., Krauter,K.S., Hewitt,J.K., 2000. Genetic and environmental influences on behavioral disinhibition. Am. J. Med. Genet. 96, 684—695.
[31]. Edenberg, H.J., Xuei, X., Chen, H.J., Tian, H., Wetherill, L.F., Dick, D.M., Almasy, L., Bierut, L., Bucholz, K.K., Goate,A., Hesselbrock, V., Kuperman, S., Nurnberger,J., Porjesz, B., Rice,J., Schuckit,M., Tischfield, J., Begleiter, H., Foroud, T., 2006. Association of alcohol dehydrogenase genes with alcohol dependence: a comprehensive analysis. Hum. Mol. Genet. 15, 1539—1549.
[32]. Kuo, P.H., Kalsi, G., Prescott, C.A., Hodgkinson, C.A., Goldman, D., van den Oord, E.J., Alexander,J., Jiang, C., Sullivan, P.F., Patterson, D.G., Walsh, D., Kendler, K.S., Riley, B.P., 2008. Association of ADHandALDH genes with alcohol dependence in the Irish Affected Sib Pair Study of alcohol dependence (IASPSAD) sample. Alcohol.: Clin. Exp. Res. 32, 785—795.
[33]. Whitfield, J.B., 1997. Meta-analysis of the effects of alcohol dehydrogenase genotype on alcohol dependence and alcoholic liver disease. Alcohol Alcohol. 32, 613—619.
[34]. Casey, B.J., Galvan, A., Hare, T.A., 2005. Changes in cerebral functional organization during cognitive development. Curr. Opin. Neurobiol. 15, 239—244.
[35]. Clark,D.B., Winters, K.C., 2002. Measuring risks and outcomes in substance use disorders prevention research. J. Consult. Clin. Psychol. 70, 1207—1223.
[36]. Vrieze, S.I., Hicks, B.M., Iacono, W.G., McGue, M., 2012. Decline in genetic influence on the co-occurrence of alcohol,marijuana, and nicotine dependence symptoms from age 14 to 29. Am. J. Psychiatry 169, 1073—1081.
[37]. Lenroot, R.K., Gogtay, N., Greenstein, D.K., Wells, E.M., Wallace, G.L., Clasen, L.S., Blumenthal, J., Lerch, J., Zijdenbos, A.P., Evans, A.C., Thompson, P.M., Giedd, J., 2007. Sexual dimorphism of brain developmental trajectories during childhood and adolescence. Neuroimage 36, 1065–1073.
[38]. Meyers, J.L., Salvatore,J.E., Vuoksimaa, E., Korhonen, T., Pulkkinen, L., Rose, R.J., Kaprio, J., Dick, D.M., 2014. Genetic influences on alcohol use behaviors have diverging developmental trajectories: a prospective study among male and female twins. Alcohol.: Clin. Exp. Res. 38, 2869–2877.
[39]. Steinberg, L., 2004. Risk taking in adolescence: what changes, and why. Ann. N. Y. Acad. Sci. 1021, 51–58.
[40]. Steinberg, L., Graham, S., O’Brien, L., Woolard, J., Cauffman, E., Banich, M., 2009. Age differences in future orientation and delay discounting. Child Dev. 80, 28–44.
[41]. Purcell, S., 2002. Variance components models for gene-environment interaction in twin analysis. Twin Res. 5, 554–571.
[42]. van der Sluis, S., Posthuma, D., Dolan, C.V., 2012. A note of false positives and power in GxE modelling of twin data. Behav. Genet. 42, 170–186.
[43]. Dick, D.M., Pagan, J.L., Viken, R., Purcell,S., Kaprio, J., Pulkkinen, L., Rose,R.J., 2007a. Changing environmental influences on substance use across development. Twin Res. Hum. Genet. 10, 315–326.
[44]. Button, T.M., Rhee, S.H., Hewitt,J.K., Young, S.E., Corley,R.P., Stallings, M.C., 2007.The role of conduct disorder in explaining the comorbidity between alcohol and illicit drug dependence in adolescence. Drug Alcohol Depend. 87, 46–53.
[45]. Feinberg, M.E., Button, T.M., Neiderhiser, J.M., Reiss, D., Hetherington, E.M., 2007.
[46]. Button, T.M., Lau, J.Y., Maughan, B., Eley, T.C., 2008. Parental punitive discipline, negative life events and gene-environment interplay in the development of externalizing behavior. Psychol. Med. 38, 29–39.
[47]. Hicks, B.M., South, S., DiRago, A.C., Iacono, W.G., McGue, M., 2009. Environmental adversity and increasing genetic risk for externalizing disorders. Arch. Gen. Psychiatry 66, 640–648.
[48]. Dick, D.M., Bernard, M., Aliev,F., Viken, R., Pulkkinen, L., Kaprio, J., Rose,R.J., 2009a. The role of socioregional factors in moderating genetic influences on early adolescent behavior problems and alcohol use. Alcohol.: Clin. Exp. Res. 33, 1739–1748.
[49]. Dick, D.M., Rose, R.J., Viken, R.J., Kaprio, J., Koskenvuo, M., 2001. Exploring gene-environment interactions: socioregional moderation of alcohol use. J. Abnorm. Psychol. 110, 625–632.
[50]. Shanahan, M.J., Hofer, S.M., 2005. Social context in gene-environment interactions: retrospect and prospect. J. Gerontol. Ser. B: Psycholo. Sci. Social Sci. 60 (Spec. No. 1), 65–76.
[51]. Kendler,K.S., Baker, J.H., 2007. Genetic influences on measures of the environment: a systematic review. Psychol. Med. 37, 615–626.
[52]. Kendler, K.S., Jacobson, K., Myers, J.M., Eaves, L.J., 2008a. A genetically informative developmental study of the relationship between conduct disorder and peer deviance in males. Psychol. Med. 38, 1001–1011.
[53]. Kendler,K.S., Jacobson, K.C., Gardner, C., Gillespie,N.A., Aggen, S.H., Prescott, C., 2007. Creating a social world: a developmental twin study of peer group deviance. Arch. Gen. Psychiatry 64, 958–965.
[54]. Dick, D.M., Agrawal, A., Keller, M.C., Adkins, A., Aliev, F., Monroe, S., Hewitt, J.K., Kendler, K.S., Sher, K.J., 2015. Candidate gene-environment interaction research: reflections and recommendations. Perspect. Psychol. Sci. 10, 37–59.
[55]. Saccone, N.L., Downey, J.T.J., Meyer, D.J., Neuman, R.J., Rice, J.P., 1999. Mapping genotype to phenotype for linkage analysis. Genet. Epidemiol. 17 (Suppl. 1), S703–S708.
[56]. Wang, S., Huang, S., Liu, N., Chen, L., Oh, C., Zhao, H., 2005. Whole-genome linkage analysis in mapping alcoholism genes using single-nucleotide polymorphisms and microsatellites. BMC Genet. 6 (Suppl. 1), S28.
[57]. Hirschhorn, J.N., Daly, M., 2005. Genome-wide association studies for common diseases and complex traits. Nat. Rev. – Genet. 6, 95–108.
[58]. Ott,J., Wang, J., Leal,S., 2015. Genetic linkage analysis in the age of whole-genome sequencing. Nat. Rev. – Genet. 16, 275–284.
[59]. Risch, N., Merikangas, K., 1996. The future of genetic studies of complex human diseases. Science 273, 1516–1517.
[60]. McCarthy, M.I., Abecasis, G.R., Cardon, L.R., Goldstein, D.B., Little, J., Ioannidis, J.P., Hirschhorn, J.N., 2008. Genome-wide association studies for complex traits: consensus, uncertainty and challenges. Nat. Rev. – Genet. 9, 356–369.
[61]. Barrett,J.C., Fry,B., Maller, J., Daly,M.J., 2005. Haploview: analysis and visualization of LD and haplotype maps. Bioinformatics 21, 263–265.









