1. Introduction
With the rapid development of science and technology, the demand for energy is increasing. However, fossil fuel reserves are limited, and over-exploitation of fossil fuels has caused serious energy shortages and environmental problems, so it is crucial to develop environmentally friendly and clean energy sources [1]. Hydrogen energy has become the focus of current research due to its advantages such as high calorific value of combustion, no environmental pollution and renewable. There are many hydrogen production schemes, among which electrolytic water hydrogen production technology is considered as one of the promising hydrogen production methods due to its advantages such as high purity of hydrogen production and non-polluting products. Currently, electrolytic water hydrogen production technology still relies on Pt-group precious metal catalysts, whose high price limits large-scale application [2]. Academician Tao Zhang proposed monoatomic catalysts, which, despite their superiority in terms of metal atom utilization and electrochemical reaction catalytic activity, inhibit oxygen adsorption activation due to their single active site and symmetric electronic structure. In order to solve this problem, researchers further synthesized diatomic catalysts, which were optimized by using the synergistic effect of diatoms. At present, there are fewer literature summaries on diatomic catalysts, and the research is still in the groping stage. In this paper, the application of catalysts in hydrogen production from electrolytic water is summarized, which has certain reference significance for the synthesis and optimization of electrocatalysts.
Hydrogen production from electrolyzed water is a new clean energy technology to prepare high-purity hydrogen and oxygen by electrolyzing water molecules. Since the discovery of the phenomenon of water electrolysis in 1780 to the development to the present, alkaline electrolysis of water dominates due to the mature technology and lower cost. The alkaline electrolyzed water hydrogen production device consists of electrolyte, electrodes, diaphragm, and electrolysis cell.
2. Principles of electrolyzed water to hydrogen technology
The basic working mechanism of hydrogen production from electrolyzed water can be summarized as a two-electrode electrochemical reaction process. When an appropriate potential is applied by the external power source, the ions in the electrolyte will migrate directionally and participate in the electrode reaction: in the cathode region, protons (H+) gather on the electrode surface under the driving force of the electric field, and hydrogen is generated by obtaining electrons in the reduction reaction. At the same time, hydroxide ions (OH-) at the interface of the anode release electrons in the oxidation, which is ultimately converted into oxygen. The whole system realizes the energy conversion process of decomposition of water molecules into hydrogen and oxygen gases through two synergistic electrochemical reactions (Hydrogen Extraction Reaction HER and Oxygen Extraction Reaction OER) [3,4].
2.1. Hydrogen precipitation reaction
During water electrolysis, the hydrogen precipitation reaction in the cathode region is mainly realized through two typical pathways: the Volmer-Heyrovsk mechanism and the Volmer-Tafel mechanism. The microscopic process of this reaction shows significant differences depending on the pH difference of the working environment, which can be divided into two different reaction modes under different conditions: acidic and alkaline media [3,4]. The reaction mechanism under acidic conditions is as follows.
Volmer Reaction: \( {H^{+}}+{e^{-}}→{H_{ad}} \) (1)
Heyrovsky Reaction: \( {H_{ad}}+{H^{+}}+{e^{-}}→{H_{2}} \) (2)
Tafel Reaction: \( {H_{ad}}+{H_{ad}}→{H_{2}} \) (3)
The reaction mechanism under alkaline conditions is
Volmer Reaction: \( {H_{2}}O+{e^{-}}→{H_{ad}}+O{H^{-}} \) (4)
Heyrovsky Reaction: \( {H_{ad}}+{H_{2}}{O^{+}}+{e^{-}}→{H_{2}}+O{H^{- }} \) (5)
2.2. Oxygen precipitation reaction
Oxygen precipitation reaction is carried out on the anode of the water electrolysis device, and the reaction mechanism can be categorized into under acidic and alkaline conditions depending on the acidity or alkalinity of the electrolyte [5]:
The reaction mechanism under alkaline conditions is
\( *+O{H^{-}}{→^{*}}OH+{e^{-}} \) (6)
\( *OH+O{H^{-}}{→^{*}}O+{H_{2}}O+{e^{-}} \) (7)
\( *O+O{H^{-}}{→^{*}}OOH+{e^{-}} \) (8)
\( *OOH+O{H^{-}}{→^{*}}{O_{2}}+{H_{2}}O+{e^{-}} \) (9)
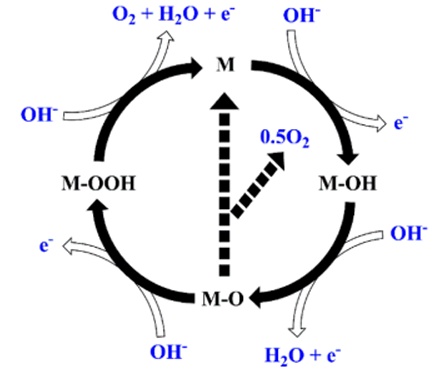
Figure 1: OER reaction mechanism under alkaline conditions
The reaction mechanism of oxygen precipitation on the surface of metal oxides in alkaline medium is shown in Fig. 1. Taking the active site M as an example, the hydroxyl group at the initiation stage of the reaction binds to the active site by chemisorption and forms M-OH intermediates by one-electron oxidation. Subsequently, it is converted to M-O species by proton-electron synergistic transfer process. There are two branching paths for the subsequent reaction: path 1 is nucleophilic attack of M-O by hydroxide ions, which generates M-OOH intermediate through electron desorption, and finally releases oxygen and restores the active site through proton-coupled electron transfer; path 1 generates oxygen molecules through direct coupling of the two M-O species, and regenerates the catalytically active site at the same time.
3. Overview and research progress of electro-catalysts
3.1. Precious metal based catalysts
Precious metal Pt-based electrocatalysts have good catalytic effect, high commercialization, and the most extensive application. Although the catalytic performance of Pt is very good, there are problems such as insufficient stability and easy deactivation in long-term operation, so the large-scale commercial application of PEMFC [6] is still hindered. In addition, due to the scarcity and high price of Pt-based metals, in order to reduce the dosage of Pt, it is more common practice to disperse the nanoparticles of Pt on activated carbon with a large specific surface area. The electrochemically active surface area of Pt/C catalysts decreases significantly after a long period of time of operation in PEMFC, and the Pt active sites are greatly reduced, resulting in poor catalytic performance [7]. The current Pt catalysts have the following deficiencies:
(1) Pt migrates and agglomerates on carbon carriers. Generally, the particle size of Pt nanoparticles is required to be 2~5 nm. however, due to the large surface energy of Pt particles with 2~5 nm particle size, they are highly prone to agglomeration. the catalytic performance of the PEMFC will be drastically reduced after a long time of operation when the Pt particles agglomerate.
(2) Pt nanoparticles will be redeposited after dissolution. During the long time running of PEMFC, the Pt catalyst will be oxidized, generating the oxides of Pt, which will be dissolved into Pt2+ and Pt4+ ions under acidic conditions, which not only affects the activity, but also causes irreversible damage to the proton exchange membrane.
(3) Pt is easily poisoned. Considering the cost and the limit of current separation and purification process, the fuel of PEMFC is not completely pure hydrogen, which contains impurities such as CO2, CO, NH3, H2S, etc., which will be strongly adsorbed on the surface of Pt catalyst, resulting in the reduction of active sites and a significant decrease in catalytic activity.
(4) Carbon carrier corrosion, Pt particles fall off and catalyst is destroyed. The fuel cell operating potential is high, under this condition, the porous carbon carrier of Pt/C catalyst used for ORR will be corroded, which leads to the separation between the Pt particles and the carrier. The Pt particles can not play a catalytic role after detaching from the three-phase interface [3].
Thus the poor stability of Pt catalysts hinders their development. Currently, the academic community generally uses methods such as crystal surface modulation, construction of polymetallic systems, surface modification, and development of non-carbon carriers in an attempt to improve the stability and catalytic activity of Pt catalysts. And at present, the low precious metal reserves and high cost limit its further development. Based on the above situation, many researchers have conducted a lot of studies on precious metal catalysts in order to be able to maintain or even enhance the reaction activity of metal catalysts even under the condition of reducing the amount of precious metals. For example, Li et al. synthesized platinum-copper (Pt/Cu) alloy catalysts in the solid-solution structure of Pt/Cu alloy in one step by solvothermal method. The atomic ratio of Pt and Cu is about 3:2 by means of morphological characterization and other tests, and the charge migration ability is enhanced, which reduces the amount of precious metal platinum and improves the HER catalytic activity of the catalyst at the same time. The HER catalytic performance of the Pt/Cu alloy showed that under the three conditions of acidic, neutral and alkaline, the Pt/Cu alloy electrocatalyst had the best performance under acidic condition, with an onset overpotential of only 20.3 mV and a Tafel slope of 37.56 mV dec-1 at a current density of 10 mA cm-2, and the impedance value of the Pt/Cu alloy was higher than that of Pt/Cu alloy with a Pt mass ratio of 20% [8]. The Pt/Cu alloy has the smallest impedance value of 28.9 Ω compared to that of Pt/C with 20% Pt mass.
3.2. Non-precious etal based catalysts
In addition to platinum, other members of the platinum family of metals exhibit electrocatalytic properties, but still face bottlenecks such as high preparation costs, limited catalytic efficiency and insufficient cycling stability in practical applications. In order to solve the above technical shortcomings, the development of non-precious metal catalyst systems needs to meet the key design indexes: the construction of new catalytic systems needs to follow the following core guidelines: (1) The catalytic efficiency and operational stability needs to reach a comparable level of performance with platinum-based catalysts. (2) electrolyte corrosion resistance, not easy to redox reactions leading to corrosion; (3) good electrical conductivity; (4) high specific surface area, capable of adequate adsorption; (5) structural stability, good durability; (6) high surface mass transfer rate.
Based on the above design principles, a transition metal-based non-precious metal catalyst (NPMC) can be used to replace the Pt cathode. NPMCs can be broadly categorized into two types: transition metal compounds and transition metal-carbon composites. This paper mainly summarizes the catalysts from three categories: transition metal carbides, transition metal nitrides, and transition metal nitrogen-carbon composites.
3.3. Transition metal nitrides
The embedding of N atoms in transition metal nitrides can expand the metal lattice and broaden the d-band of the metal by hybridizing with the s and p orbitals of the adjacent atoms. The electronic structure of the transition metal nitrides can be converged to the characteristics of noble metals by modulating the contraction effect of the center of the d-band and optimizing the distribution of the density of states near the Fermi energy level. This directional reconstruction of the electronic structure effectively regulates the electronic states on the catalyst surface, and ultimately significantly enhances its intrinsic catalytic activity. For example, Liu et al [9] who utilized Fe doping to change the electronic structure of Ni3N to alter the adsorption strength of ions and promote the electrolytic water reaction. The Fe-doped Ni3N (Fe-Ni3N/NF) synthesized in situ on the surface of nickel foam by using nickel foam as a carrier through hydrothermal reaction combined with Ar/NH3 post-treatment. By means of SEM and TEM characterization, it was found that Fe-Ni3N showed irregular particles, and a short dendritic structure consisting of irregular particles with good electrocatalytic performance was formed on NF, and the overpotential of Fe-Ni3N/NF was only 124 mV at a current density of 10 mA cm-2, and the Tafel slope was 69 mV dec-1, indicating that the reaction kinetics was also good.
3.4. Transition metal nitrogen-carbon compounds
Since the above transition metal compounds only exhibit good ORR catalytic activity in alkaline environments, their performance is severely degraded in acidic environments, and the catalysts corrode, dissolve, or oxidize at higher operating potentials, with a narrow range of applicability [6]. In contrast, transition metal-carbon composites are more stable and exhibit good ORR catalytic activity in both acid and alkaline environments. Among them, transition metal-nitrogen-carbon (M-NC) composites are considered as the most promising non-precious metal catalysts for oxygen reduction reaction to replace Pt-based catalysts because of their high catalytic activity, stability, environmental friendliness, low price, and durability. The catalytic mechanism of the current M-NC catalysts is still unclear, and it is generally believed that defective carbon and metal centers are the catalytic active sites of M-NC. The introduction of hard/flexible templates can be used to increase the specific surface area of the catalyst and thus enhance the catalytic performance, and common templates include silica spheres and mesoporous silicon [5]. For the preparation of M-NC catalysts, the pyrolysis method is mostly used. The pyrolysis method can be divided into in situ pyrolysis and loading method, in which the in situ pyrolysis method is also called one-step pyrolysis method, i.e., only through a one-step reaction, high temperature pyrolysis of the precursor, to realize the in situ doping, and to obtain the catalysts with good performance [8,9]. This method has been widely studied because of its simple steps, high reproducibility, and favorable for large-scale production and application. However, in the pyrolysis process, metal thermal reduction reaction, heteroatom doping reaction, graphite carbonization reaction, etc. are involved. These reactions in turn interact with each other. During the thermal reduction process, metal nanoparticles are precipitated, and these precipitated metal nanoparticles catalyze the graphitization reaction of the carbon layer, which leads to the fact that metal particles are usually encapsulated in the outer graphitic carbon layer during the high-temperature preparation of M-NC catalysts. A series of complex reactions interact with each other, resulting in poor controllability of the high-temperature pyrolysis process, and the structure and morphology of the catalysts obtained will be different. For these reasons, the design concept and the means of regulating the preparation of catalysts for the oxygen reduction reaction by pyrolysis are of paramount importance [11]. There are two main types of precursors using in situ pyrolysis: metal-organic frameworks (MOFs) systems, and metal inorganic salt systems [10]. MOFs compounds are diverse, with a large specific surface area and pore structure, possessing a high degree of designability and tailoring, and are generally connected by ligand bonds between the central metal atoms and the organic ligands, which is favorable for the generation of the M-Nx structure and contributes to the ORR active center. In the M-Nx system, MOFs compounds such as ZIF, Prussian blue and MIL are generally used as precursors.
In different MOFs, factors such as the types of central metal atoms and ligands, the type of coordination between them, and the assembly pattern affect the degree and size of metal nanoparticles precipitated during pyrolysis, which in turn affects the ORR activity. In recent years, there have also been studies using nitrogen-containing small molecules (dicyandiamide, melamine) as precursors, but compared with them, nitrogen-containing polymers have a regular structure and good thermal stability, and can still maintain a high nitrogen content after high-temperature treatment [11]. Composite carrier method, template method and encapsulation method are commonly used. Composite carriers are often selected as a combination of carbon nanoparticles, carbon nanotubes, graphene, etc., whose composite structure can provide mesoporous channels for catalysts to improve mass transfer and oxygen reduction activity; while in the template method, one is to use silica, mesoporous silica, etc., which are stable at high temperatures and can be removed by pyrolysis as templates. However, there are many reactions involved in high temperature environments, and in order to obtain a stable, uniform, and controllable nanoencapsulated structure, it is necessary to rationally select the metal center, nitrogen source and carbon sources. The encapsulation method is generally not applied alone, but often used in synergy with the template method to prepare electrocatalysts with homogeneous structure and abundant active sites [12]. The loading method, also called loaded doping preparation method, uses carbon material as a carrier, doped with heteroatoms and metal atoms, and then prepared by pyrolysis to obtain catalysts. Among the carbon materials can be commercially available carbon black, carbon nanotubes, graphene, etc. or pre-prepared mesoporous carbon, bioporous carbon, etc.
The catalysts prepared by the loading method, in which the carbon has been molded long ago, are not greatly affected by pyrolysis on their morphology, and their catalytic activity relies on the interaction and synergistic effect between the dopant atoms and the functional groups on the carbon surface. Although the current M-NC catalysts have shown catalytic activity not weaker than that of Pt/C catalysts, there are still many difficulties to further improve their performance. It is currently believed that the catalytic activity and stability of M-NC catalysts depend on two factors: first, the number and distribution of active sites. The type of dopant atoms and the interactions between dopant atoms will affect the active sites and thus the catalytic activity. Transition metal elements play an extremely important role in M-NC catalysts. Among them, iron and cobalt possess the best ORR catalytic activity. Catalysts doped with both iron and cobalt atoms will have even better catalytic activity. In addition, the doping of nitrogen atoms also has a significant effect on the ORR catalytic performance of the materials. Nitrogen atoms can directly participate in the catalytic process of ORR as well as coordinate the environment, and at the same time, they can complex with transition metal protons to generate the M-Nx structure, which contributes to the center of ligand bonding catalytic activity. The second is the specific surface area and pore structure of the material. These two factors not only significantly affect the number and distribution of active sites on the surface of M-NC catalysts, but also play a decisive role in the adsorption rate and mass transfer rate when electrochemical reactions occur.
4. Conclusion and outlook
The core of hydrogen production technology from electrolyzed water lies in the development of efficient, stable and low-cost electrocatalysts. This paper summarizes the research progress on the improvement strategies of noble metal-based catalysts (e.g., crystal surface modulation, multi-alloying) as well as non-precious metal-based catalysts (transition metal carbides, nitrides and M-NC materials). The synergistic effect of diatomic catalysts and the optimization of electronic structure of transition metal-based materials have significantly enhanced the catalytic activity, and the performance of some non-precious metal catalysts in alkaline medium has been close to that of Pt-based materials. However, the existing studies still face the following two challenges:
(1) balance between catalytic activity and stability: noble metal catalysts need to further inhibit particle agglomeration and corrosion, and the performance of non-precious metal materials in acidic media still needs a breakthrough.
(2) Precise regulation of active sites: the mechanism of the active center of M-NC catalysts is still unclear, and the design needs to be optimized by combining theoretical calculations and experimental verification.
References
[1]. WANG S M, ZHANG X F, KUANG F H, et al. Preparation and properties of a new porous ceramic material used in cleanenergy field[J]. Journal of Materials Science & Technology, 2(7): 1255 − 1260.
[2]. HAMK, BAES, LEE J. Classification and technical target of water electrolysis for hydrogen production[J]. Journal of Energy Chemistry, 2024, 95: 554 − 576.
[3]. TAK S S, SHETYE O, MULEY O, et al. Emerging technologies for hydrogen production from wastewater[J]. International Journal of Hydrogen Energy, 2022, 47(88): 37282 − 37301.
[4]. XU X X, ZHOU Q, YU D H. The future of hydrogen energy: Biohydrogen production technology[J]. International Journal of Hydrogen Energy, 2022, 47(79): 33677 − 33698.
[5]. ZHENG B, GAO T F, SUN P, et al. Effects of solidparticle aspect ratio on the performance of hydrogen production by electrolyzed water[J]. International Journal of Hydrogen Energy, 2023, 48(19): 7038 −7047.
[6]. DOS SANTOS K G, ECKERT C T, DE ROSSI E, et al.Hydrogen production in the electrolysis of water in Brazil, a review[J]. Renewable and Sustainable Energy Reviews, 2017, 68:563 − 571.
[7]. GAO D H, JI S, WANG H, et al. Amorphous co-modified catalyst based on interfacial coupling effect for efficient hydrogen production by water electrolysis[J]. Journal of Alloys and Compounds, 2023, 960: 170832.
[8]. LIAO P S, KANG J W, ZHONG Y C, et al. Recent advances of two-dimensional metal-organic frameworks in alkaline electrolysis water for hydrogen production[J]. Science China Chemistry, 2023, 66 (7): 1924 − 1939.
[9]. LIU Q, YANG S, LIU Y H, et al. F-doped self-supportingcarbon electrode to improve carbon–water reaction assisted electrolysis of water for hydrogen production[J]. Fuel, 2024, 375:132512.
[10]. WANG H Z, GAO Q, SUN S F, et al. CoP@NRGO composite as a high-efficiency water electrolysis catalyst for hydrogen generation[J]. Journal of Solid State Chemistry, 2020, 290:121596.
[11]. ZHAN T J, BIE R S, SHEN Q H, et al. Application ofelectrolysis water hydrogen production in the field of renewable energy power generation[J]. IOP Conference Series: Earth and Environmental Science, 2020, 598 (1): 012088.
[12]. XUE F M, SU J C, LI P P, et al. Application of proton exchangemembrane electrolysis of water hydrogen production technology in power plant[J]. IOP Conference Series: Earth and Environmental Science, 2021, 631 (1): 012079.
Cite this article
Su,J. (2025). Progress in the Application of Electrocatalysts. Applied and Computational Engineering,143,47-53.
Data availability
The datasets used and/or analyzed during the current study will be available from the authors upon reasonable request.
Disclaimer/Publisher's Note
The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of EWA Publishing and/or the editor(s). EWA Publishing and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content.
About volume
Volume title: Proceedings of the 3rd International Conference on Functional Materials and Civil Engineering
© 2024 by the author(s). Licensee EWA Publishing, Oxford, UK. This article is an open access article distributed under the terms and
conditions of the Creative Commons Attribution (CC BY) license. Authors who
publish this series agree to the following terms:
1. Authors retain copyright and grant the series right of first publication with the work simultaneously licensed under a Creative Commons
Attribution License that allows others to share the work with an acknowledgment of the work's authorship and initial publication in this
series.
2. Authors are able to enter into separate, additional contractual arrangements for the non-exclusive distribution of the series's published
version of the work (e.g., post it to an institutional repository or publish it in a book), with an acknowledgment of its initial
publication in this series.
3. Authors are permitted and encouraged to post their work online (e.g., in institutional repositories or on their website) prior to and
during the submission process, as it can lead to productive exchanges, as well as earlier and greater citation of published work (See
Open access policy for details).
References
[1]. WANG S M, ZHANG X F, KUANG F H, et al. Preparation and properties of a new porous ceramic material used in cleanenergy field[J]. Journal of Materials Science & Technology, 2(7): 1255 − 1260.
[2]. HAMK, BAES, LEE J. Classification and technical target of water electrolysis for hydrogen production[J]. Journal of Energy Chemistry, 2024, 95: 554 − 576.
[3]. TAK S S, SHETYE O, MULEY O, et al. Emerging technologies for hydrogen production from wastewater[J]. International Journal of Hydrogen Energy, 2022, 47(88): 37282 − 37301.
[4]. XU X X, ZHOU Q, YU D H. The future of hydrogen energy: Biohydrogen production technology[J]. International Journal of Hydrogen Energy, 2022, 47(79): 33677 − 33698.
[5]. ZHENG B, GAO T F, SUN P, et al. Effects of solidparticle aspect ratio on the performance of hydrogen production by electrolyzed water[J]. International Journal of Hydrogen Energy, 2023, 48(19): 7038 −7047.
[6]. DOS SANTOS K G, ECKERT C T, DE ROSSI E, et al.Hydrogen production in the electrolysis of water in Brazil, a review[J]. Renewable and Sustainable Energy Reviews, 2017, 68:563 − 571.
[7]. GAO D H, JI S, WANG H, et al. Amorphous co-modified catalyst based on interfacial coupling effect for efficient hydrogen production by water electrolysis[J]. Journal of Alloys and Compounds, 2023, 960: 170832.
[8]. LIAO P S, KANG J W, ZHONG Y C, et al. Recent advances of two-dimensional metal-organic frameworks in alkaline electrolysis water for hydrogen production[J]. Science China Chemistry, 2023, 66 (7): 1924 − 1939.
[9]. LIU Q, YANG S, LIU Y H, et al. F-doped self-supportingcarbon electrode to improve carbon–water reaction assisted electrolysis of water for hydrogen production[J]. Fuel, 2024, 375:132512.
[10]. WANG H Z, GAO Q, SUN S F, et al. CoP@NRGO composite as a high-efficiency water electrolysis catalyst for hydrogen generation[J]. Journal of Solid State Chemistry, 2020, 290:121596.
[11]. ZHAN T J, BIE R S, SHEN Q H, et al. Application ofelectrolysis water hydrogen production in the field of renewable energy power generation[J]. IOP Conference Series: Earth and Environmental Science, 2020, 598 (1): 012088.
[12]. XUE F M, SU J C, LI P P, et al. Application of proton exchangemembrane electrolysis of water hydrogen production technology in power plant[J]. IOP Conference Series: Earth and Environmental Science, 2021, 631 (1): 012079.









