1. Introduction
Psychological disorders are defined by the disrupted behaviours and mental patterns that deviates an individual from normal functioning; they are some of the hardest to treat diseases. Many psychological disorders are difficult to treat due to our lack of understanding of their causes and mechanisms of action. Furthermore, mental illnesses can be challenging to diagnose since the line between a “normal response” and “mentally ill” is often ambiguous. This paper discusses about the disorder, schizophrenia, and two existing treatments for it. The current understanding of schizophrenia is limited. However, studies are supporting that the disorder is caused by a combination of heredity and abnormal functioning of neurotransmitters [1,2]. The treatments for schizophrenia—antipsychotics—helped to elucidate the disorder’s mechanism of action better by examining the effects of antipsychotics on different receptors and binding partners. Antipsychotics are categorized as typical or atypical, both types are successful in reducing schizophrenia symptoms in patients, while the atypical drugs are evidently stronger [3]. However, all antipsychotics have a variety of side effects, indicating that better treatments may be needed moving forwards.
2. Overview of Schizophrenia
2.1. Symptoms of Schizophrenia
Schizophrenia is a chronic mental disorder characterized by the alteration in perceptions, beliefs and consciousness and the disconnection with reality [4]. Patients with schizophrenia may experience symptoms characterized by positive, negative, and cognitive symptoms. Positive symptoms describe the excesses in behaviour, which include hallucinations (false perception), delusions (false personal beliefs), thought disorder, and movement disorder; negative symptoms are associated with deficits in apathy, avolition, and blunted emotion; cognitive symptoms include impairments in executive function, attention, and memory [5]. Antipsychotics mainly target positive symptoms, while negative symptoms are more challenging to treat.
2.2. Impact of the Disease
Schizophrenia affects around 0.7-1% of the population [6]. A meta-review was conducted with a 90% confidence interval to show that individuals affected by schizophrenia are likely to have a high standardized mortality ratio of 2.5 [7]. The reduction in life expectancy associated with schizophrenia is about 10-20 years (compared with heavy smoking: 8-10 years), and the employment rate is as low as 10-20% [7,8].
2.3. Causes of Schizophrenia
The diathesis-stress model proposes that the symptoms of schizophrenia are triggered by psychosocial stress acting on preexisting vulnerable genetic disposition. In other words, the cause of schizophrenia is due to the combination of, or the interaction between genetic predisposition and the environment. The genetic factor contributes to the disease substantially. The risk of having schizophrenia for monozygotic twins is 45.6%, and up to 13.7% for first-degree relatives—a family member (sibling, one’s offspring or parent) whose genetic information is about 50% identical to an individual [1]. Monozygotic twins have identical genes; if the genetic factor contributes solely to the disease, the twins will have a concordance of 100%. This emphasizes the effect of environmental factors, as well as the genetic component.
Furthermore, multiple studies indicate that multiple genes contribute to schizophrenia, making the disease polygenic. The phenotype heterogeneity (the wide variety of symptoms) of schizophrenia is believed to be associated with genetic heterogeneity, meaning a genetic disorder or a phenotype is caused by multiple alleles [9]. This hypothesis views each gene as a factor, along with environmental factors, contributing to the schizophrenia phenotypes. Each factor contributes towards a critical threshold at which symptoms will manifest. However, the degree of contribution of the genes remains unknown as the specific genes associated with schizophrenia still have not been identified.
Nevertheless, multiple studies attempted to identify the genes that cause the high heritability of schizophrenia. Linkage study is based on how recombination, or crossing over, occurs in homologous chromosomes during meiosis. Gene loci located closer together are more likely to be inherited together after recombination. In contrast, the probability that one gene is crossed over to the homologous chromosome increases when the two genes are farther away from each other [9]. If a schizophrenia gene is located next (linked) to a marker gene, then the probability of finding the disease causing gene increases (Figure 1). A recent study used a genome scan meta-analysis (GSMA) on genome-wide linkage scans of schizophrenia and found multiple schizophrenia candidate genes [11]. Chromosomes have two arms; the shorter arm is referred to as “p” (in petite), and the longer arm is denoted as “q”. The group discovered linkage on chromosomes 1q, 2q, 5q, and 8p. In addition, a more recent study discovered a linkage signal on the 22q11 chromosome, the gene which encodes for proline dehydrogenase, also known as proline oxidase [12]. Overexpression of gene 22p11 can cause interference with the metabolism of the amino acid L-proline, in turn affecting the synthesis of glutamate. This neurotransmitter may be one of the causes of schizophrenia [12,13].
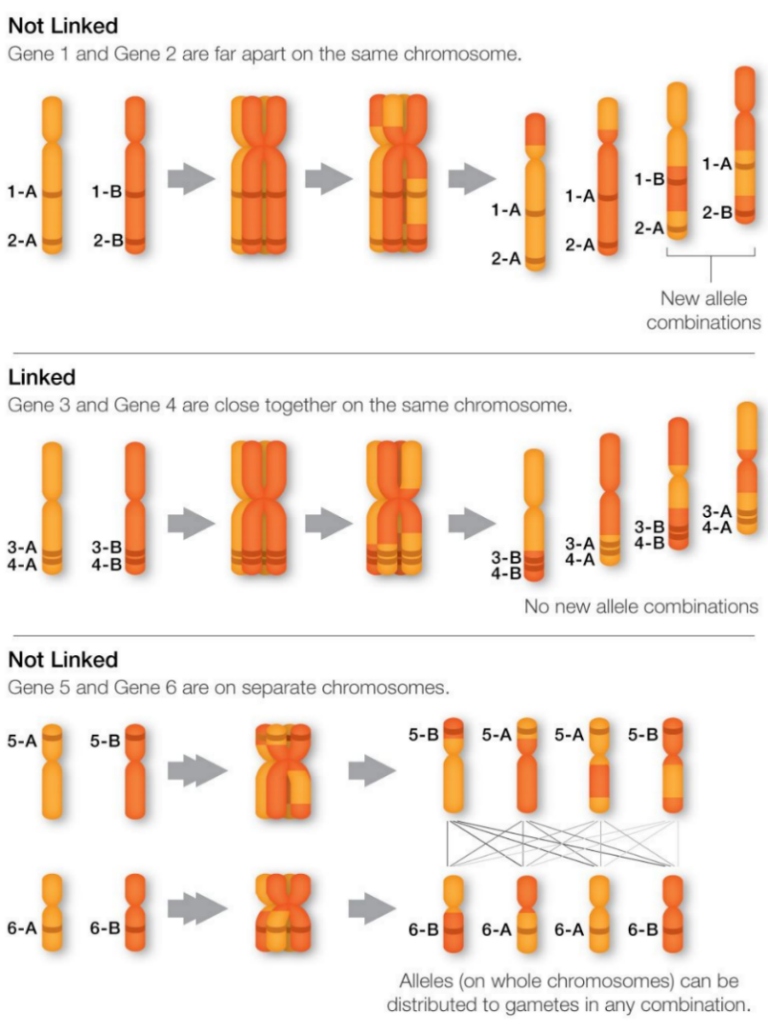
Figure 1. Linkage gene versus non-linkage genes during crossover in meiosis [10].
The catechol-O-methyltransferase (COMT) gene, also located on the 22q11 chromosome, was also studied. COMT is an enzyme that degrades catecholamines, including dopamine, by methylation [14]. Dopamine is a critical neurotransmitter that is hypothesized to be involved in a pathway that causes schizophrenia. The abnormal activity of the enzyme affects the prefrontal lobe, which is responsible for executive function; dysfunction of the prefrontal lobe is also associated with the disease [15]. Individuals with schizophrenia tend to have a lower prefrontal lobe volume. Additionally, the enzyme’s low activity causes failure to break down the increased dopamine, thereby increasing the risk of developing schizophrenia.
In addition, cases where individuals develop schizophrenia without the influence of genetics (non-familial), are sporadic. These individuals may be phenocopies, in which the schizophrenia phenotypes are caused by psychosocial causes, drug misuse, or brain trauma, such as epilepsies [9].
3. Treatment for Schizophrenia
3.1. Typical vs. Atypical Antipsychotics
Antipsychotics (APDs) are psychiatric drugs that are prescribed to treat psychosis and psychosis-related symptoms, including schizophrenia, bipolar disorder, major depressive disorder, personality disorder, Etc [16].
Typical APDs, or first-generation APDs, are often known for their ability to treat the positive symptoms of schizophrenia [17]. They are also popular regarding their serious side effects, such as extrapyramidal side effects, tardive dyskinesia, metabolic syndromes, Parkinsonism, and potentially fatal conditions, such as neuroleptic malignant syndrome [18]. The cause of the motor impairment side effects is believed to be the drug’s high affinity to the dopamine D2 receptors. All APDs block dopamine D2 receptors, while typical APDs have a higher affinity to the receptors, meaning they bind more tightly to the receptors [17].
It was believed that typical and atypical APDs solely differ in their affinity to dopamine D2 receptors [19]. Atypical APDs indeed have less affinity to the dopamine D2 receptors, so they dissociate from the receptor quicker [17]. However, atypical APDs also target multiple other receptors—which is discussed below—on postsynaptic neurons (Figure 2).
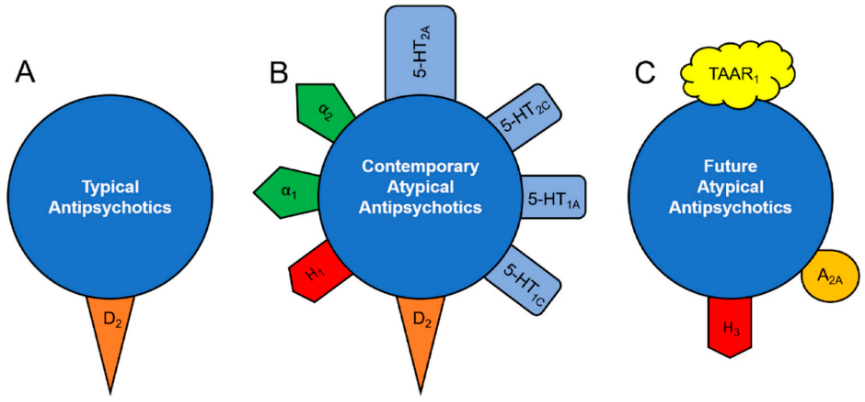
Figure 2. The receptors that different types of antipsychotics target [20].
Even though the adverse side effects of the first-generation APDs, they can still be found as first-line treatment for psychosis because they are relatively inexpensive [18]. Two APDs, haloperidol and clozapine, are chosen to be discussed herein.
3.1.1. The Discovery and Synthesis of Haloperidol
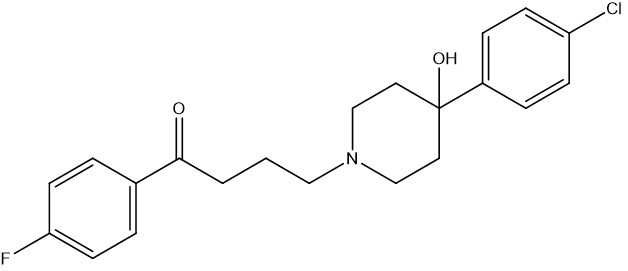
Figure 3. The chemical structure of haloperidol
Haloperidol, or 4-[4-(4-chlorophenyl)-4-hydroxypiperidin-1-yl]-1-(4-fluorophenyl)butan-1-one, is a butyrophenone derivative discovered in the 1950s by Dr. Paul A. J. Janssen [22]. Janssen was known for discovering dextromoramide, an analgesic with a brief duration of action [23]. After that discovery, he began to do research on pethidine (or meperidine), he obtained compound R951 (Figure 5) by the Mannich reaction of acetophenone with normeperidine [22,23]. Mannich reaction (Figure 4) is similar to dehydrative condensation, where a bond is formed between the reactants and water, is produced as the byproduct [26]. He then lengthened the carbon chain to produce R1187.
After numerous trials and errors, the compound R1625 (Figure 5, 6), haloperidol, was discovered as a potent tranquillizer in 1958 [22]. The compound mimics chlorpromazine, the first typical APD discovered, but was 50-100 times stronger than chlorpromazine.
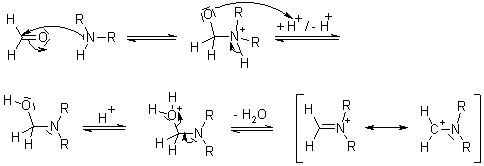
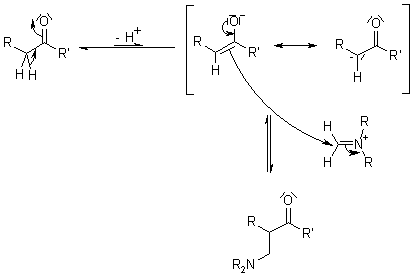
Figure 4. Mannich reaction mechanism shown by the reaction between an amine with formaldehyde, and carbonyl compound with an α acidic hydrogen [24].
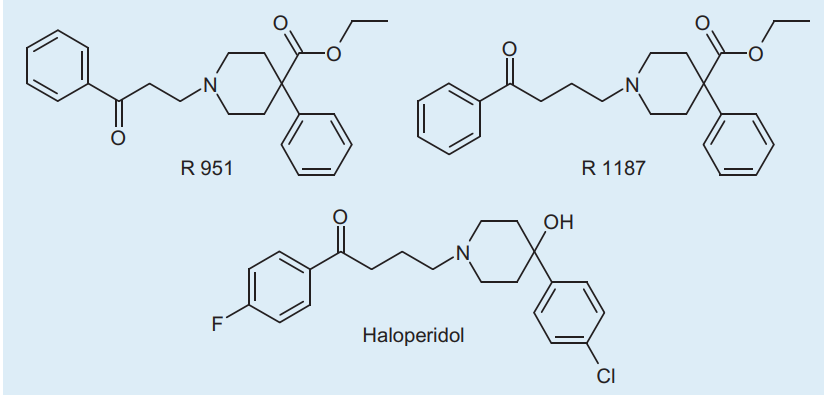
Figure 5. The important compounds Jenssen produced during his research about pethidine and piperidine [25]
3.1.2. Pharmacology of Haloperidol.
Haloperidol can be taken either orally, intravenously, or via intramuscular injection. Haloperidol is widely known for its antagonism of dopamine D2 receptors. In addition, the drug also binds to 5-HT2A (serotonin-2A) receptors, α1A (adrenergic) receptors, and α1B receptors [26]. Haloperidol is highly selective for dopamine D2 receptors because it has a high affinity for them—about 100 times higher than clozapine [26]. All the receptors it affects are G-Protein Coupled Receptors (GCPR) involved in the functioning of neuronal pathways [27,28]. G-Protein coupled receptors are proteins with seven transmembrane domains that receives extracellular signals and translates them into intracellular signals [29]. G proteins are specialized proteins that can bind with GTP or GDP. When bound to GTP, G protein subunits are activated and can perform activities signalled by the extracellular receptor. Haloperidol may be affecting these receptors in the mesolimbic pathway, but no significant evidence was found [30].
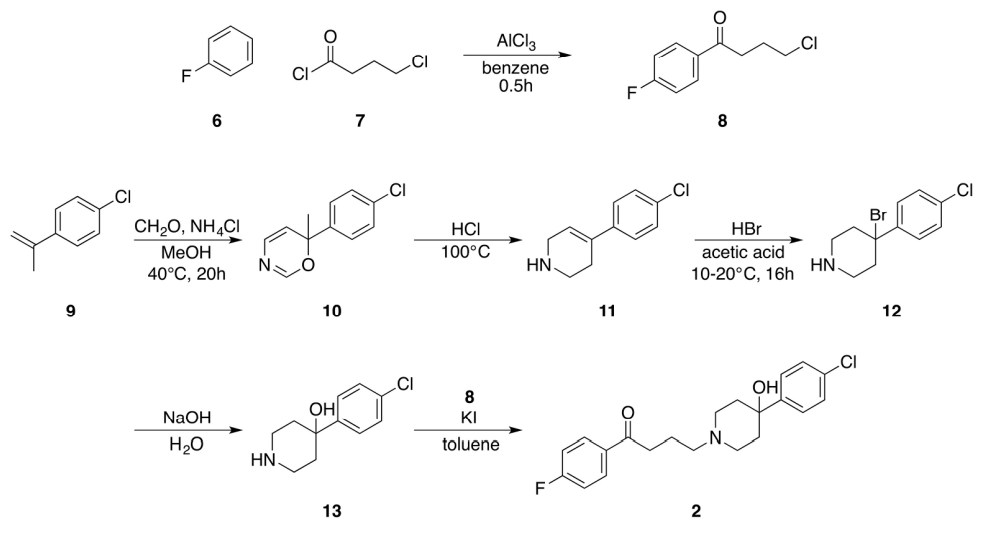
Figure 6. The original synthesis of haloperidol by Jenssen Pharmaceutica [25].
Although the definite mechanism of APDs has not yet been discovered, dozens of in vivo studies identified the blockage of striatal dopamine D2 receptor when patients have been treated with typical APDs [31]. The striatum is a critical component of the basal ganglia (Figure 7) in our brain. Those parts of the brain are apart of the nigrostriatal pathway, which is responsible for motor control [2]. Therefore, the nigrostriatal pathway may as well be responsible for the extrapyramidal side effects [32].
3.1.3. Side Effects of Haloperidol
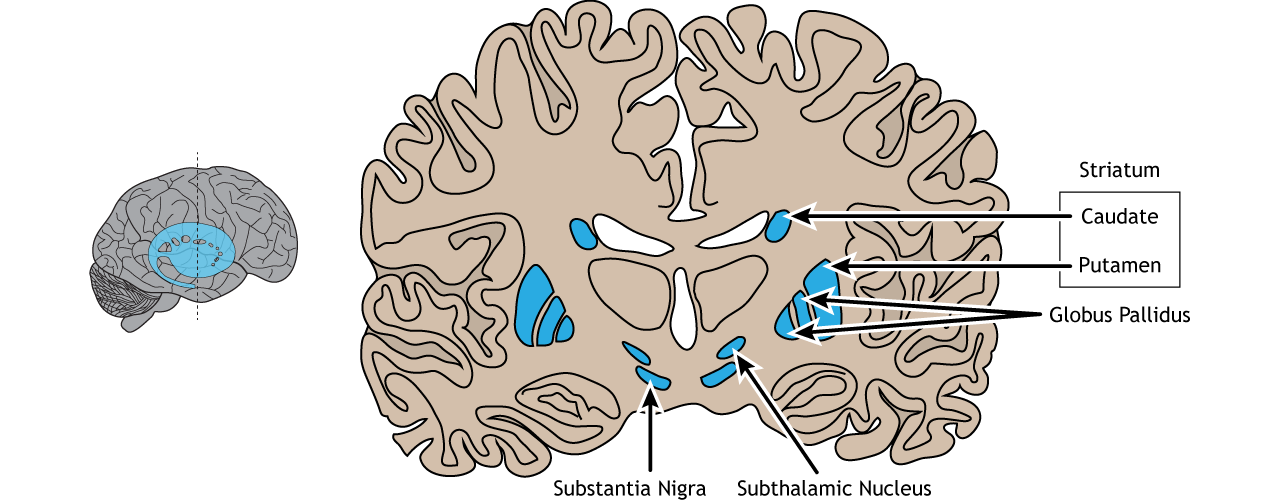
Figure 7. The location of the components of basal ganglia. The striatum is composed by the caudate and the putamen [35].
The basal ganglia use three different pathways to ensure the performance of the correct movement. The three pathways are the direct, indirect, and hyperdirect pathway [33]. This paper will only focus on the direct and indirect pathway (Figure 8). The direct pathway involves the striatum receiving excitatory information from the glutamatergic neurons from the cerebral cortex; then, the striatum sends signals through inhibitory neurons: GABAergic neurons to the internal globus pallidus (GPi) and the reticular substantia nigra (SNr). Information is relayed at the thalamus and sent back to the cortex by excitatory neurons [33]. In other words, the cortex sends excitatory signals (glutamate) to the striatum, stimulating it to send inhibitory signals (GABA) to the GPi. This suppresses the GPi to send inhibitory signals to the thalamus, leading the thalamus to release excitatory signals to the cortex, causing movement and triggering positive feedback.
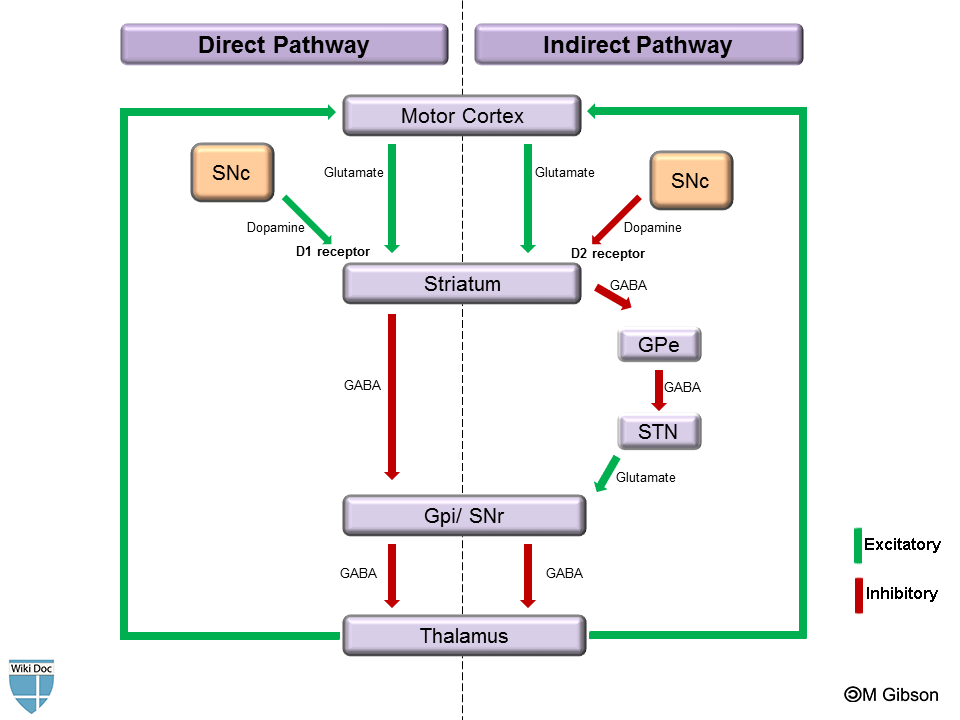
Figure 8. A diagram of the direct and indirect pathways [34]. SN stands for substantia nigra. SN is further divided into the reticular part (SNr), and the compact part (SNc). The globus pallidus is shortened as GP. GPe and GPi refers to the external and internal part. STN is the subthalamus nucleus.
In addition, the SNc sends dopamine to the striatal dopamine D2 receptors via the indirect pathway (Figure 8). Dopamine excites the release of GABA to the GPe, inhibiting the release of GPe’s GABA to the subthalamus nucleus (STN). The excitatory output from the STN to GPi increases inhibition of the thalamus, decreasing excitatory output to the cortex, thereby decreasing unwanted movement [35]. Thus, haloperidol blocks the binding of dopamine to D2 receptors inhibits the release of GABA to GPe. Through cascade effect, the thalamus is not inhibited, leading it to send excitatory signals to the cortex, triggering unwanted movements [2,36]
3.1.4. The Discovery and Synthesis of Clozapine
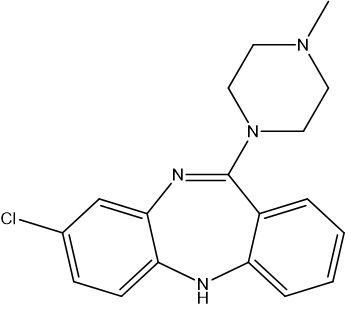
Figure 9. Chemical structure of clozapine
Clozapine (8-chloro-11-(4-methylpiperazin-1-yl)-5H-dibenzo[b,e][1,4]diazepine) is a tricyclic benzodiazepine drug synthesized in the 1960s by a health care company called Sandoz (now known as Novartis) [38]. The first step of synthesis involves an Ullmann reaction between aryl halides 10 and 11 (Figure 10) catalyzed by copper [39]. A reduction of the nitro moiety of 12, then refluxing the product in dimethylbenzene will result in the core structure 14. POCl3 provides the molecule with a better leaving group, chloride, to react with N-methylpiperazine to form clozapine 3 [38].
The drug is known for its efficacy in treating treatment-resistant schizophrenia patients and its low rates of causing extrapyramidal side effects [40]. A study in 1988 conducted a clinical trial with 286 patients who were unresponsive to haloperidol after 6 weeks of treatment [3]. Haloperidol nonresponders were randomly assigned to be treated with chlorpromazine or clozapine for 6 weeks. It was found 30% of clozapine-treated patients improved, while only 4% of chlorpromazine-treated patients were considered responders. Furthermore, typical APDs require ~75% striatal D2 receptors occupancy effectiveness (haloperidol induces ~80% occupancy), while clozapine displays only <60% occupancy [36,38]. Thus, dopamine D2 receptors must not be the only type of receptor which clozapine targets. In addition, the low D2 occupancy may explain clozapine’s lower propensity for extrapyramidal side effects.
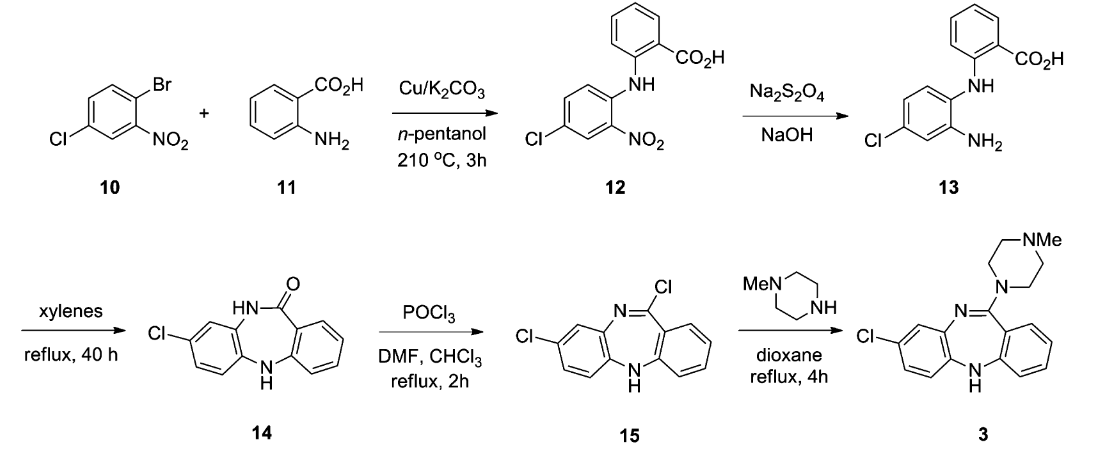
Figure 10. The original synthesis steps of clozapine [38]. Compound 14, 8-Chloro-5,10-dihydro-11H-dibenzo[b,e][1,4]-diazepin-11-one, is the core structure of clozapine, which can be synthesized in various ways.
3.1.5. The Polypharmacology of Clozapine
Clozapine is administered orally; 12.5 mg initially and increase by 25-50 mg to 300-450 mg over a two-week period if no side effects are observed [40].
Meltzer (1989) was the first to propose the serotonin-dopamine hypothesis for schizophrenia [41]. Clozapine acts on both the serotonin (5-HT) and the dopamine systems because it is a potent blocker of the 5-HT2A receptors and a weaker antagonist of dopamine D2 receptors [41]. Clozapine also blocks 5-HT agonists, such as quipazine and MK-212 [41]. MK-212 binds to the 5-HT2A receptor and stimulates the secretion of cortisol and prolactin in human [42]. Another study reported that clozapine is an inverse agonist for 5-HT2A [43]. The 5-HT2A receptor is also a GPCR located on postsynaptic neurons, requiring a ligand for activation. An antagonist only blocks the action of the ligand but does not alter the basal activity (the ability for the receptor to be activated) of the receptor [44]. An inverse agonist will lower the basal activity. This may increase presynaptic dopamine levels in nigrostriatal neurons, further decreasing the risk of developing Parkinsonism and other extrapyramidal side effects. Additionally, Meyer and Stahl reported that the inverse agonism decreases dopamine in the mesolimbic system, which is responsible for its antipsychotic effect mentioned earlier [43].
Positron emission tomography (PET) studies show that basal ganglia D2 receptor occupancy of clozapine is <50%. Cortical 5-HT2A occupancy is as high as 90% for clozapine and other atypical APDs, including risperidone and olanzapine [45].
However, as shown in Figure 2, the drug also shows considerable affinities to multiple other receptors, such as 5-HTreceptors subtypes, D1, D4, α1-adrenergic, α2-adrenergic, H1 histaminergic, and M1 cholinergic receptors [45]. This leads to the question of which pharmacological property is responsible for the drug’s therapeutic efficacy.
3.1.6. Side Effects Clozapine.
Clozapine may induce neutropenia and severe neutropenia (also known as agranulocytosis in old literature) [46]. Severe neutropenia occurs when the absolute neutrophil count is <500/μl. In 1975, 0.7% of clozapine patients in Finland developed severe neutropenia, and 50% of these patients died from secondary infections [47]. Thus, patients treated with clozapine must undergo hematological monitoring of their neutrophil count. A study discussed the pharmacogenetic factor of clozapine-induced neutropenia [48]. The study suggested that patients with lower genetic risk may be less likely to develop neutropenia, thus can relax the monitoring.
Furthermore, metabolic syndrome, such as weight gain and type II diabetes, induced by clozapine is also a source of distress for patients. The antagonism of 5-HT2C receptors can cause obesity in mice, which could also contribute to weight gain in humans [49]. 5-HT2C indirectly acts on the hypothalamus, a structure that is involved in the central control of hunger and feeding [50]. Appetite is suppressed by 5-HT signalling on pro-opiomelanocortin (POMC) neurons within the hypothalamus. Treatment for obesity often includes medications that are 5-HT agonists to induce an anorexigenic effect. Additionally, clozapine can cause orthostatic hypotension, seizures, and myocarditis, observed in 3% of schizophrenia patients [51]. Severe cases of myocarditis may show respiratory and cardiovascular symptoms.
4. Conclusion
The definite mechanism of both haloperidol and clozapine (in fact, all APDs) is yet to be understood, just as the cause of schizophrenia. However, evidence shows that haloperidol works on dopamine D2 receptors, leading researchers to hypothesize the mesolimbic pathway and dopamine hypothesis. Likewise, the polypharmacology of clozapine leads researchers to propose the serotonin-dopamine hypothesis [41]. From this, we can deduce that the belief that typical APDs and atypical APDs solely differ in their affinity to D2 receptors is incorrect. Clozapine is concluded to be superior to haloperidol in treating both positive and negative symptoms and more severe and lethal in the side effects it causes [52]. In addition, most psychological disorders are caused by genetic disposition and the environment. It is likely that schizophrenia is a polygenic disorder, such that multiple genes contribute to the manifestation of the symptoms. In future experiments, studies could potentially be done on the polygenicity aspect of schizophrenia. Whole-genome sequencing applications could help identify all disease-causing alleles so we could attempt to understand their biological mechanisms [53].
References
[1]. Fowles, D. C. (1992). Schizophrenia: Diathesis-stress revisited. Annual Review of Psychology, 43(1), 303–336. https://doi.org/10.1146/annurev.ps.43.020192.001511
[2]. Meltzer, H. Y., & Stahl, S. M. (1976). The dopamine hypothesis of schizophrenia: A review*. Schizophrenia Bulletin, 2(1), 19–76. https://doi.org/10.1093/schbul/2.1.19
[3]. Kane, J., Honigfield, G., Singer, J., & Meltzer, H. (1988). Clozapine for the treatment-resistant schizophrenic. Archives of General Psychiatry, 45(9), 789. https://doi.org/10.1001/ archpsyc.1988.01800330013001
[4]. Patel, K. R., Cherian, J., Gohil, K., & Atkinson, D. (2014, September). Schizophrenia: Overview and treatment options. P & T : a peer-reviewed journal for formulary management. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4159061/
[5]. Owen, M. J., Sawa, A., & Mortensen, P. B. (2016). Schizophrenia. The Lancet, 388(10039), 86–97. https://doi.org/10.1016/s0140-6736(15)01121-6
[6]. Howrigan, D. P., Rose, S. A., Samocha, K. E., Fromer, M., Cerrato, F., Chen, W. J., Churchhouse, C., Chambert, K., Chandler, S. D., Daly, M. J., Dumont, A., Genovese, G., Hwu, H.-G., Laird, N., Kosmicki, J. A., Moran, J. L., Roe, C., Singh, T., Wang, S.-H., … Neale, B. M. (2020). Exome sequencing in schizophrenia-affected parent–offspring trios reveals risk conferred by protein-coding de novo mutations. Nature Neuroscience, 23(2), 185–193. https://doi.org/10.1038/s41593-019-0564-3
[7]. Chesney, E., Goodwin, G. M., & Fazel, S. (2014). Risks of all-cause and suicide mortality in mental disorders: A Meta-Review. World Psychiatry, 13(2), 153–160. https://doi.org/10.1002/wps.20128
[8]. Marwaha, S., & Johnson, S. (2004). Schizophrenia and employment. Social Psychiatry and Psychiatric Epidemiology, 39(5), 337–349. https://doi.org/10.1007/s00127-004-0762-4
[9]. Tsuang, M. T., Stone, W. S., & Faraone, S. V. (2018). Genes, environment and schizophrenia. British Journal of Psychiatry, 178(S40). https://doi.org/10.1192/bjp.178.40.s18
[10]. Mahbuba, S. (2022, November 13). Linkage and recombination (part 1): Chromosomal theory, Linkage & crossing over. Plantlet. https://plantlet.org/linkage-and-recombination-part-1-chromosomal-theory-linkage-crossing-over/
[11]. Ng, M. Y., Levinson, D. F., Faraone, S. V., Suarez, B. K., DeLisi, L. E., Arinami, T., Riley, B., Paunio, T., Pulver, A. E., Irmansyah, Holmans, P. A., Escamilla, M., Wildenauer, D. B., Williams, N. M., Laurent, C., Mowry, B. J., Brzustowicz, L. M., Maziade, M., Sklar, P., … Lewis, C. M. (2008). Meta-analysis of 32 genome-wide linkage studies of schizophrenia. Molecular Psychiatry, 14(8), 774–785. https://doi.org/10.1038/mp.2008.135
[12]. Trifu, S., Kohn, B., Vlasie, A., & Patrichi, B.-E. (2020). Genetics of schizophrenia (review). Experimental and Therapeutic Medicine. https://doi.org/10.3892/etm.2020.8973
[13]. Cappelletti, P., Tallarita, E., Rabattoni, V., Campomenosi, P., Sacchi, S., & Pollegioni, L. (2018). Proline oxidase controls proline, glutamate, and glutamine cellular concentrations in a U87 glioblastoma cell line. PLOS ONE, 13(4). https://doi.org/10.1371/journal.pone.0196283
[14]. Forster, P. (2015, November 8). Same - S-adenosyl-methionine. Gateway Psychiatric. https://www.gatewaypsychiatric.com/same-s-adenosyl-methionine/
[15]. Weinberger, D. R., Aloia, M. S., Goldberg, T. E., & Berman, K. F. (1994). The frontal lobes and schizophrenia. The Journal of neuropsychiatry and clinical neurosciences, 6(4), 419–427. https://doi.org/10.1176/jnp.6.4.419
[16]. Chokhawala, K., & Stevens, L. (2023, February 26). Antipsychotic medications - statpearls - NCBI bookshelf. National Library of Medicine. https://www.ncbi.nlm.nih.gov/ books/NBK519503/
[17]. Mathews, M., & Muzina, D. J. (2007). Atypical antipsychotics: New Drugs, new challenges. Cleveland Clinic Journal of Medicine, 74(8), 597–606. https://doi.org/10.3949/ccjm.74.8.597
[18]. Saha, K. B., Bo, L., Zhao, S., Xia, J., Sampson, S., & Zaman, R. U. (2016). Chlorpromazine versus atypical antipsychotic drugs for schizophrenia. Cochrane Database of Systematic Reviews, 2016(4). https://doi.org/10.1002/14651858.cd010631.pub2
[19]. Meltzer, H. Y., & Gadaleta, E. (2021). Contrasting typical and atypical antipsychotic drugs. FOCUS, 19(1), 3–13. https://doi.org/10.1176/appi.focus.20200051
[20]. Grinchii, D., & Dremencov, E. (2020). Mechanism of action of atypical antipsychotic drugs in mood disorders. International Journal of Molecular Sciences, 21(24), 9532. https://doi.org/10.3390/ijms21249532
[21]. Haloperidol. National Center for Biotechnology Information. PubChem Compound Database. (n.d.). https://pubchem.ncbi.nlm.nih.gov/compound/haloperidol#section=3D-Conformer
[22]. Raviña, E., & Kubinyi, H. (2011). The evolution of drug discovery from traditional medicines to modern drugs. Wiley-VCH-Verl.
[23]. Sneader, W. (2006). Drug discovery: A history. John Wiley & Sons. U’S. (2022, August 1). Mannich reaction mechanism - reaction details and applications. BYJUS. https://byjus.com/chemistry/mannich-reaction-mechanism/
[24]. Mannich reaction. Organic Chemistry. (n.d.). https://www.organic-chemistry.org/ namedreactions/mannich-reaction.shtm
[25]. Wermuth, C. G., Villoutreix, B., Grisoni, S., Olivier, A., & Rocher, J.-P. (2015). Strategies in the search for new lead compounds or original working hypotheses. The Practice of Medicinal Chemistry, 73–99. https://doi.org/10.1016/b978-0-12-417205-0.00004-3
[26]. Tyler, M. W., Zaldivar-Diez, J., & Haggarty, S. J. (2017). Classics in chemical neuroscience: Haloperidol. ACS Chemical Neuroscience, 8(3), 444–453. https://doi.org/10.1021/ acschemneuro.7b00018
[27]. Perez, D. M. (2020). Α1-adrenergic receptors in neurotransmission, synaptic plasticity, and cognition. Frontiers in Pharmacology, 11. https://doi.org/10.3389/fphar.2020.581098
[28]. Kasai, R. S., Ito, S. V., Awane, R. M., Fujiwara, T. K., & Kusumi, A. (2017). The class-A GPCR dopamine D2 receptor forms transient dimers stabilized by agonists: Detection by single-molecule tracking. Cell Biochemistry and Biophysics, 76(1–2), 29–37. https://doi.org/10.1007/s12013-017-0829-y
[29]. Wacker, D., Stevens, R. C., & Roth, B. L. (2017). How ligands illuminate GPCR molecular pharmacology. Cell, 170(3), 414–427. https://doi.org/10.1016/j.cell.2017.07.009
[30]. Mackay, A. V., Iversen, L. L., Rossor, M., Spokes, E., Bird, E., Arregui, A., Creese, I., & Synder, S. H. (1982). Increased brain dopamine and dopamine receptors in schizophrenia. Archives of General Psychiatry, 39(9), 991–997. https://doi.org/10.1001/archpsyc.1982.04290090001001
[31]. Agid, O., Mamo, D., Ginovart, N., Vitcu, I., Wilson, A. A., Zipursky, R. B., & Kapur, S. (2006). Striatal vs extrastriatal dopamine D2 receptors in antipsychotic response—a double-blind pet study in Schizophrenia. Neuropsychopharmacology, 32(6), 1209–1215. https://doi.org/ 10.1038/sj.npp.1301242
[32]. D’Souza, R. S., & Hooten, W. M. (2023). Extrapyramidal symptoms - StatPearls - NCBI BOOKSHELF. National Library of Medicine. https://www.ncbi.nlm.nih.gov/ books/NBK534115/
[33]. Andrușca , A. (2023, July 27). Basal ganglia pathways. Kenhub. https://www.kenhub.com/ en/library/anatomy/direct-and-indirect-pathways-of-the-basal-ganglia
[34]. Gibson, C. M. (n.d.). Basal ganglia. wikidoc. https://www.wikidoc.org/index.php/ Basal_gangliaman, M. (2020). Know your brain: Striatum. @neurochallenged. https://neuroscientificallychallenged.com/posts/know-your-brain-striatum
[35]. Henley, C. (2021, January 1). Basal ganglia. Foundations of Neuroscience. https://openbooks.lib.msu.edu/neuroscience/chapter/basal-ganglia/
[36]. Xiberas, X., Martinot, J. L., Mallet, L., Artiges, E., Loc’h, C., Mazière, B., & Paillère-Martinot, M. L. (2001). Extrastriatal and striatal D2 dopamine receptor blockade with haloperidol or new antipsychotic drugs in patients with schizophrenia. British Journal of Psychiatry, 179(6), 503–508. https://doi.org/10.1192/bjp.179.6.503
[37]. U.S. National Library of Medicine. (n.d.). Clozapine. National Center for Biotechnology Information. PubChem Compound Database. https://pubchem.ncbi.nlm.nih.gov/ compound/clozapine
[38]. Wenthur, C. J., & Lindsley, C. W. (2013). Classics in chemical neuroscience: Clozapine. ACS Chemical Neuroscience, 4(7), 1018–1025. https://doi.org/10.1021/cn400121z
[39]. Yang, Q., Zhao, Y., & Ma, D. (2022). CU-mediated Ullmann-type cross-coupling and industrial applications in route design, process development, and scale-up of pharmaceutical and Agrochemical Processes. Organic Process Research & Development, 26(6), 1690–1750. https://doi.org/10.1021/acs.oprd.2c00050
[40]. Clozapine monograph for professionals. Drugs.com. (2021). https://www.drugs.com/ monograph/clozapine.html
[41]. Meltzer, H. Y. (1989). Clinical studies on the mechanism of action of clozapine: The dopamine-serotonin hypothesis of schizophrenia. Psychopharmacology, 99(S1). https://doi.org/10.1007/ bf00442554
[42]. Lowy, M. T., & Meltzer, H. Y. (1988). Stimulation of serum cortisol and prolactin secretion in humans by MK-212, a centrally active serotonin agonist. Biological Psychiatry, 23(8), 818–828. https://doi.org/10.1016/0006-3223(88)90070-4
[43]. Meyer, J. M., & Stahl, S. M. (2020). Binding Profile of Clozapine and Its Primary Active Metabolite Norclozapine. In The clozapine handbook: Stahl’s Handbooks (pp. 101–105). essay, Cambridge University Press.
[44]. Gavriilidou, A. F., Hunziker, H., Mayer, D., Vuckovic, Z., Veprintsev, D. B., & Zenobi, R. (2018). Insights into the basal activity and activation mechanism of the β1 adrenergic receptor using native mass spectrometry. Journal of the American Society for Mass Spectrometry, 30(3), 529–537. https://doi.org/10.1007/s13361-018-2110-z
[45]. Kuroki, T., Nagao, N., & Nakahara, T. (2008). Neuropharmacology of second-generation antipsychotic drugs: A validity of the serotonin–dopamine hypothesis. Progress in Brain Research, 199–212. https://doi.org/10.1016/s0079-6123(08)00910-2
[46]. Nucifora, F. C., Mihaljevic, M., Lee, B. J., & Sawa, A. (2017). Clozapine as a model for antipsychotic development. Neurotherapeutics, 14(3), 750–761. https://doi.org/10.1007/s13311-017-0552-9
[47]. Atkin, K., Kendall, F., Gould, D., Freeman, H., Lieberman, J., & O’Sullivan, D. (1996). Neutropenia and agranulocytosis in patients receiving clozapine in the UK and Ireland. British Journal of Psychiatry, 169(4), 483–488. https://doi.org/10.1192/bjp.169.4.483
[48]. Li, K. J., Solomon, H. V., & DeLisi, L. E. (2018). Clozapine pharmacogenomics. Current Opinion in Psychiatry, 31(5), 403–408. https://doi.org/10.1097/yco.0000000000000440
[49]. Reynolds, G. P., Zhang, Z.-J., & Zhang, X.-B. (2002). Association of antipsychotic druginduced weight gain with a 5-HT2C receptor gene polymorphism. The Lancet, 359(9323), 2086–2087. https://doi.org/10.1016/s0140-6736(02)08913-4
[50]. Przegaliński, E., Witek, K., Wydra, K., Kotlińska, J. H., & Filip, M. (2023). 5-HT2C receptor stimulation in obesity treatment: Orthosteric agonists vs. Allosteric modulators. Nutrients, 15(6), 1449. https://doi.org/10.3390/nu15061449
[51]. Haidary, H. A., & Padhy, R. K. (2021). Clozapine - statpearls - NCBI bookshelf. National Library of Medicine. https://www.ncbi.nlm.nih.gov/books/NBK535399/
[52]. Breier, A., Buchanan, R. W., Kirkpatrick, B., Davis, O. R., Irish, D., Summerfelt, A., & Carpenter, W. T. (1994). Effects of clozapine on positive and negative symptoms in outpatients with schizophrenia. American Journal of Psychiatry, 151(1), 20–26. https://doi.org/10.1176/ajp.151.1.20
[53]. Owen, M. J., Legge, S. E., Rees, E., Walters, J. T., & O’Donovan, M. C. (2023). Genomic findings in schizophrenia and their implications. Molecular Psychiatry. https://doi.org/10.1038/ s41380-023-02293-8
Cite this article
Liao,B. (2024). Comparing haloperidol with clozapine as treatments for schizophrenia. Theoretical and Natural Science,45,9-20.
Data availability
The datasets used and/or analyzed during the current study will be available from the authors upon reasonable request.
Disclaimer/Publisher's Note
The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of EWA Publishing and/or the editor(s). EWA Publishing and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content.
About volume
Volume title: Proceedings of the 2nd International Conference on Modern Medicine and Global Health
© 2024 by the author(s). Licensee EWA Publishing, Oxford, UK. This article is an open access article distributed under the terms and
conditions of the Creative Commons Attribution (CC BY) license. Authors who
publish this series agree to the following terms:
1. Authors retain copyright and grant the series right of first publication with the work simultaneously licensed under a Creative Commons
Attribution License that allows others to share the work with an acknowledgment of the work's authorship and initial publication in this
series.
2. Authors are able to enter into separate, additional contractual arrangements for the non-exclusive distribution of the series's published
version of the work (e.g., post it to an institutional repository or publish it in a book), with an acknowledgment of its initial
publication in this series.
3. Authors are permitted and encouraged to post their work online (e.g., in institutional repositories or on their website) prior to and
during the submission process, as it can lead to productive exchanges, as well as earlier and greater citation of published work (See
Open access policy for details).
References
[1]. Fowles, D. C. (1992). Schizophrenia: Diathesis-stress revisited. Annual Review of Psychology, 43(1), 303–336. https://doi.org/10.1146/annurev.ps.43.020192.001511
[2]. Meltzer, H. Y., & Stahl, S. M. (1976). The dopamine hypothesis of schizophrenia: A review*. Schizophrenia Bulletin, 2(1), 19–76. https://doi.org/10.1093/schbul/2.1.19
[3]. Kane, J., Honigfield, G., Singer, J., & Meltzer, H. (1988). Clozapine for the treatment-resistant schizophrenic. Archives of General Psychiatry, 45(9), 789. https://doi.org/10.1001/ archpsyc.1988.01800330013001
[4]. Patel, K. R., Cherian, J., Gohil, K., & Atkinson, D. (2014, September). Schizophrenia: Overview and treatment options. P & T : a peer-reviewed journal for formulary management. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4159061/
[5]. Owen, M. J., Sawa, A., & Mortensen, P. B. (2016). Schizophrenia. The Lancet, 388(10039), 86–97. https://doi.org/10.1016/s0140-6736(15)01121-6
[6]. Howrigan, D. P., Rose, S. A., Samocha, K. E., Fromer, M., Cerrato, F., Chen, W. J., Churchhouse, C., Chambert, K., Chandler, S. D., Daly, M. J., Dumont, A., Genovese, G., Hwu, H.-G., Laird, N., Kosmicki, J. A., Moran, J. L., Roe, C., Singh, T., Wang, S.-H., … Neale, B. M. (2020). Exome sequencing in schizophrenia-affected parent–offspring trios reveals risk conferred by protein-coding de novo mutations. Nature Neuroscience, 23(2), 185–193. https://doi.org/10.1038/s41593-019-0564-3
[7]. Chesney, E., Goodwin, G. M., & Fazel, S. (2014). Risks of all-cause and suicide mortality in mental disorders: A Meta-Review. World Psychiatry, 13(2), 153–160. https://doi.org/10.1002/wps.20128
[8]. Marwaha, S., & Johnson, S. (2004). Schizophrenia and employment. Social Psychiatry and Psychiatric Epidemiology, 39(5), 337–349. https://doi.org/10.1007/s00127-004-0762-4
[9]. Tsuang, M. T., Stone, W. S., & Faraone, S. V. (2018). Genes, environment and schizophrenia. British Journal of Psychiatry, 178(S40). https://doi.org/10.1192/bjp.178.40.s18
[10]. Mahbuba, S. (2022, November 13). Linkage and recombination (part 1): Chromosomal theory, Linkage & crossing over. Plantlet. https://plantlet.org/linkage-and-recombination-part-1-chromosomal-theory-linkage-crossing-over/
[11]. Ng, M. Y., Levinson, D. F., Faraone, S. V., Suarez, B. K., DeLisi, L. E., Arinami, T., Riley, B., Paunio, T., Pulver, A. E., Irmansyah, Holmans, P. A., Escamilla, M., Wildenauer, D. B., Williams, N. M., Laurent, C., Mowry, B. J., Brzustowicz, L. M., Maziade, M., Sklar, P., … Lewis, C. M. (2008). Meta-analysis of 32 genome-wide linkage studies of schizophrenia. Molecular Psychiatry, 14(8), 774–785. https://doi.org/10.1038/mp.2008.135
[12]. Trifu, S., Kohn, B., Vlasie, A., & Patrichi, B.-E. (2020). Genetics of schizophrenia (review). Experimental and Therapeutic Medicine. https://doi.org/10.3892/etm.2020.8973
[13]. Cappelletti, P., Tallarita, E., Rabattoni, V., Campomenosi, P., Sacchi, S., & Pollegioni, L. (2018). Proline oxidase controls proline, glutamate, and glutamine cellular concentrations in a U87 glioblastoma cell line. PLOS ONE, 13(4). https://doi.org/10.1371/journal.pone.0196283
[14]. Forster, P. (2015, November 8). Same - S-adenosyl-methionine. Gateway Psychiatric. https://www.gatewaypsychiatric.com/same-s-adenosyl-methionine/
[15]. Weinberger, D. R., Aloia, M. S., Goldberg, T. E., & Berman, K. F. (1994). The frontal lobes and schizophrenia. The Journal of neuropsychiatry and clinical neurosciences, 6(4), 419–427. https://doi.org/10.1176/jnp.6.4.419
[16]. Chokhawala, K., & Stevens, L. (2023, February 26). Antipsychotic medications - statpearls - NCBI bookshelf. National Library of Medicine. https://www.ncbi.nlm.nih.gov/ books/NBK519503/
[17]. Mathews, M., & Muzina, D. J. (2007). Atypical antipsychotics: New Drugs, new challenges. Cleveland Clinic Journal of Medicine, 74(8), 597–606. https://doi.org/10.3949/ccjm.74.8.597
[18]. Saha, K. B., Bo, L., Zhao, S., Xia, J., Sampson, S., & Zaman, R. U. (2016). Chlorpromazine versus atypical antipsychotic drugs for schizophrenia. Cochrane Database of Systematic Reviews, 2016(4). https://doi.org/10.1002/14651858.cd010631.pub2
[19]. Meltzer, H. Y., & Gadaleta, E. (2021). Contrasting typical and atypical antipsychotic drugs. FOCUS, 19(1), 3–13. https://doi.org/10.1176/appi.focus.20200051
[20]. Grinchii, D., & Dremencov, E. (2020). Mechanism of action of atypical antipsychotic drugs in mood disorders. International Journal of Molecular Sciences, 21(24), 9532. https://doi.org/10.3390/ijms21249532
[21]. Haloperidol. National Center for Biotechnology Information. PubChem Compound Database. (n.d.). https://pubchem.ncbi.nlm.nih.gov/compound/haloperidol#section=3D-Conformer
[22]. Raviña, E., & Kubinyi, H. (2011). The evolution of drug discovery from traditional medicines to modern drugs. Wiley-VCH-Verl.
[23]. Sneader, W. (2006). Drug discovery: A history. John Wiley & Sons. U’S. (2022, August 1). Mannich reaction mechanism - reaction details and applications. BYJUS. https://byjus.com/chemistry/mannich-reaction-mechanism/
[24]. Mannich reaction. Organic Chemistry. (n.d.). https://www.organic-chemistry.org/ namedreactions/mannich-reaction.shtm
[25]. Wermuth, C. G., Villoutreix, B., Grisoni, S., Olivier, A., & Rocher, J.-P. (2015). Strategies in the search for new lead compounds or original working hypotheses. The Practice of Medicinal Chemistry, 73–99. https://doi.org/10.1016/b978-0-12-417205-0.00004-3
[26]. Tyler, M. W., Zaldivar-Diez, J., & Haggarty, S. J. (2017). Classics in chemical neuroscience: Haloperidol. ACS Chemical Neuroscience, 8(3), 444–453. https://doi.org/10.1021/ acschemneuro.7b00018
[27]. Perez, D. M. (2020). Α1-adrenergic receptors in neurotransmission, synaptic plasticity, and cognition. Frontiers in Pharmacology, 11. https://doi.org/10.3389/fphar.2020.581098
[28]. Kasai, R. S., Ito, S. V., Awane, R. M., Fujiwara, T. K., & Kusumi, A. (2017). The class-A GPCR dopamine D2 receptor forms transient dimers stabilized by agonists: Detection by single-molecule tracking. Cell Biochemistry and Biophysics, 76(1–2), 29–37. https://doi.org/10.1007/s12013-017-0829-y
[29]. Wacker, D., Stevens, R. C., & Roth, B. L. (2017). How ligands illuminate GPCR molecular pharmacology. Cell, 170(3), 414–427. https://doi.org/10.1016/j.cell.2017.07.009
[30]. Mackay, A. V., Iversen, L. L., Rossor, M., Spokes, E., Bird, E., Arregui, A., Creese, I., & Synder, S. H. (1982). Increased brain dopamine and dopamine receptors in schizophrenia. Archives of General Psychiatry, 39(9), 991–997. https://doi.org/10.1001/archpsyc.1982.04290090001001
[31]. Agid, O., Mamo, D., Ginovart, N., Vitcu, I., Wilson, A. A., Zipursky, R. B., & Kapur, S. (2006). Striatal vs extrastriatal dopamine D2 receptors in antipsychotic response—a double-blind pet study in Schizophrenia. Neuropsychopharmacology, 32(6), 1209–1215. https://doi.org/ 10.1038/sj.npp.1301242
[32]. D’Souza, R. S., & Hooten, W. M. (2023). Extrapyramidal symptoms - StatPearls - NCBI BOOKSHELF. National Library of Medicine. https://www.ncbi.nlm.nih.gov/ books/NBK534115/
[33]. Andrușca , A. (2023, July 27). Basal ganglia pathways. Kenhub. https://www.kenhub.com/ en/library/anatomy/direct-and-indirect-pathways-of-the-basal-ganglia
[34]. Gibson, C. M. (n.d.). Basal ganglia. wikidoc. https://www.wikidoc.org/index.php/ Basal_gangliaman, M. (2020). Know your brain: Striatum. @neurochallenged. https://neuroscientificallychallenged.com/posts/know-your-brain-striatum
[35]. Henley, C. (2021, January 1). Basal ganglia. Foundations of Neuroscience. https://openbooks.lib.msu.edu/neuroscience/chapter/basal-ganglia/
[36]. Xiberas, X., Martinot, J. L., Mallet, L., Artiges, E., Loc’h, C., Mazière, B., & Paillère-Martinot, M. L. (2001). Extrastriatal and striatal D2 dopamine receptor blockade with haloperidol or new antipsychotic drugs in patients with schizophrenia. British Journal of Psychiatry, 179(6), 503–508. https://doi.org/10.1192/bjp.179.6.503
[37]. U.S. National Library of Medicine. (n.d.). Clozapine. National Center for Biotechnology Information. PubChem Compound Database. https://pubchem.ncbi.nlm.nih.gov/ compound/clozapine
[38]. Wenthur, C. J., & Lindsley, C. W. (2013). Classics in chemical neuroscience: Clozapine. ACS Chemical Neuroscience, 4(7), 1018–1025. https://doi.org/10.1021/cn400121z
[39]. Yang, Q., Zhao, Y., & Ma, D. (2022). CU-mediated Ullmann-type cross-coupling and industrial applications in route design, process development, and scale-up of pharmaceutical and Agrochemical Processes. Organic Process Research & Development, 26(6), 1690–1750. https://doi.org/10.1021/acs.oprd.2c00050
[40]. Clozapine monograph for professionals. Drugs.com. (2021). https://www.drugs.com/ monograph/clozapine.html
[41]. Meltzer, H. Y. (1989). Clinical studies on the mechanism of action of clozapine: The dopamine-serotonin hypothesis of schizophrenia. Psychopharmacology, 99(S1). https://doi.org/10.1007/ bf00442554
[42]. Lowy, M. T., & Meltzer, H. Y. (1988). Stimulation of serum cortisol and prolactin secretion in humans by MK-212, a centrally active serotonin agonist. Biological Psychiatry, 23(8), 818–828. https://doi.org/10.1016/0006-3223(88)90070-4
[43]. Meyer, J. M., & Stahl, S. M. (2020). Binding Profile of Clozapine and Its Primary Active Metabolite Norclozapine. In The clozapine handbook: Stahl’s Handbooks (pp. 101–105). essay, Cambridge University Press.
[44]. Gavriilidou, A. F., Hunziker, H., Mayer, D., Vuckovic, Z., Veprintsev, D. B., & Zenobi, R. (2018). Insights into the basal activity and activation mechanism of the β1 adrenergic receptor using native mass spectrometry. Journal of the American Society for Mass Spectrometry, 30(3), 529–537. https://doi.org/10.1007/s13361-018-2110-z
[45]. Kuroki, T., Nagao, N., & Nakahara, T. (2008). Neuropharmacology of second-generation antipsychotic drugs: A validity of the serotonin–dopamine hypothesis. Progress in Brain Research, 199–212. https://doi.org/10.1016/s0079-6123(08)00910-2
[46]. Nucifora, F. C., Mihaljevic, M., Lee, B. J., & Sawa, A. (2017). Clozapine as a model for antipsychotic development. Neurotherapeutics, 14(3), 750–761. https://doi.org/10.1007/s13311-017-0552-9
[47]. Atkin, K., Kendall, F., Gould, D., Freeman, H., Lieberman, J., & O’Sullivan, D. (1996). Neutropenia and agranulocytosis in patients receiving clozapine in the UK and Ireland. British Journal of Psychiatry, 169(4), 483–488. https://doi.org/10.1192/bjp.169.4.483
[48]. Li, K. J., Solomon, H. V., & DeLisi, L. E. (2018). Clozapine pharmacogenomics. Current Opinion in Psychiatry, 31(5), 403–408. https://doi.org/10.1097/yco.0000000000000440
[49]. Reynolds, G. P., Zhang, Z.-J., & Zhang, X.-B. (2002). Association of antipsychotic druginduced weight gain with a 5-HT2C receptor gene polymorphism. The Lancet, 359(9323), 2086–2087. https://doi.org/10.1016/s0140-6736(02)08913-4
[50]. Przegaliński, E., Witek, K., Wydra, K., Kotlińska, J. H., & Filip, M. (2023). 5-HT2C receptor stimulation in obesity treatment: Orthosteric agonists vs. Allosteric modulators. Nutrients, 15(6), 1449. https://doi.org/10.3390/nu15061449
[51]. Haidary, H. A., & Padhy, R. K. (2021). Clozapine - statpearls - NCBI bookshelf. National Library of Medicine. https://www.ncbi.nlm.nih.gov/books/NBK535399/
[52]. Breier, A., Buchanan, R. W., Kirkpatrick, B., Davis, O. R., Irish, D., Summerfelt, A., & Carpenter, W. T. (1994). Effects of clozapine on positive and negative symptoms in outpatients with schizophrenia. American Journal of Psychiatry, 151(1), 20–26. https://doi.org/10.1176/ajp.151.1.20
[53]. Owen, M. J., Legge, S. E., Rees, E., Walters, J. T., & O’Donovan, M. C. (2023). Genomic findings in schizophrenia and their implications. Molecular Psychiatry. https://doi.org/10.1038/ s41380-023-02293-8









